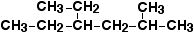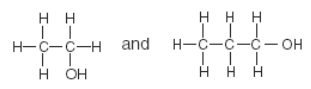|
|
WebAssigns
Sample Problems: (use the Ctrl + F feature to find your similar problem)
Answers in GREEN
Work in RED
Problem incorrect or not here? Post in the BATS FORUM
Answers in GREEN
Work in RED
Problem incorrect or not here? Post in the BATS FORUM
Intro to WebAssign Part 2
WebAssign allows you to submit your homework, quizzes, or tests online. It works from any computer with a connection to the Internet and a recent version of the most common browsers. To better understand how to use WebAssign, please answer the following questions.
(a) If an assignment covers material that has not been addressed in class, what should you do?
check with your instructor
(b) Who is responsible for setting or changing the due date of your assignment?
your teacher or someone designated by your teacher
(c) Who makes your assignments in WebAssign?
your teacher
(d) How can you tell how many submissions are allowed for each question part?
Click Show Details in the header of the question.
(e) To protect your answers after completing an assignment, what should you do?
click the logout link at the top of any WebAssign page
(f) Why should you always start a session with WebAssign by going to /login.html and logging in?
If you skip the login page, WebAssign won't know who is taking the assignment.
(g) Why is it a bad idea to reload a page?
Depending on how you arrived at the page, reloading or refreshing may resubmit your assignment and use up one of your submission allowances.
(h) Which of the following MUST be enabled on your browser?
Java or JavaScript
(i) Because the Internet is not 100% reliable,
None of these statements is correct.
2)
Answering questions in WebAssign
(a) Multiple Choice Who was the first president of the United States?
George Washington
(b) Multiple Choice with Pull-Down Menu This year belongs to the third millennium.
(c) True/False A baker's dozen is more than 12.
true
(d) Multiple Selection Which of the following cities are state capitals?
Atlanta, GA
Albany, NY
Sacramento, CA
(e) Fill-in-the-Blank Which planet is commonly referred to as the "red" planet? (Hint: Who is the god of war in Roman mythology?)
Mars
(f) Numerical What is one divided by three (1/3)?
.33
(f) Symbolic Write a formula for the area of a circle with radius r. Use r for the radius.
A = pi*r^2
(h) Essay Give three reasons for attending this class.
cool
fun
awesome
Entering numbers in WebAssign
(a) What is the minimum number of significant figures generally expected for a WebAssign answer?
three
(b) Sometimes the proper number of significant figures will be required for your answer, not just three. In this case, you will see the significant figure icon just before a question that checks for the correct number of significant figures. For example, give the number, 143.488, to 2 significant figures.
140
(c) Enter the charge of an electron (1.6 10-19 C) using the scientific "E" notation so that the number can be read by WebAssign.
1.6e-19 C
Just replace the " * 10^ " with a " E "
"times ten to the... " and "E" are the same thing.
You're just bringing the exponent down and taking away the ten, the E separates the portions of the number just like *10^ did
(d) Multiply 3.7 and 0.280 and enter the answer according to WebAssign's 1% tolerance or three-significant-figures rule for numerical answers.
3.7*.280
(a) If an assignment covers material that has not been addressed in class, what should you do?
check with your instructor
(b) Who is responsible for setting or changing the due date of your assignment?
your teacher or someone designated by your teacher
(c) Who makes your assignments in WebAssign?
your teacher
(d) How can you tell how many submissions are allowed for each question part?
Click Show Details in the header of the question.
(e) To protect your answers after completing an assignment, what should you do?
click the logout link at the top of any WebAssign page
(f) Why should you always start a session with WebAssign by going to /login.html and logging in?
If you skip the login page, WebAssign won't know who is taking the assignment.
(g) Why is it a bad idea to reload a page?
Depending on how you arrived at the page, reloading or refreshing may resubmit your assignment and use up one of your submission allowances.
(h) Which of the following MUST be enabled on your browser?
Java or JavaScript
(i) Because the Internet is not 100% reliable,
None of these statements is correct.
2)
Answering questions in WebAssign
(a) Multiple Choice Who was the first president of the United States?
George Washington
(b) Multiple Choice with Pull-Down Menu This year belongs to the third millennium.
(c) True/False A baker's dozen is more than 12.
true
(d) Multiple Selection Which of the following cities are state capitals?
Atlanta, GA
Albany, NY
Sacramento, CA
(e) Fill-in-the-Blank Which planet is commonly referred to as the "red" planet? (Hint: Who is the god of war in Roman mythology?)
Mars
(f) Numerical What is one divided by three (1/3)?
.33
(f) Symbolic Write a formula for the area of a circle with radius r. Use r for the radius.
A = pi*r^2
(h) Essay Give three reasons for attending this class.
cool
fun
awesome
Entering numbers in WebAssign
(a) What is the minimum number of significant figures generally expected for a WebAssign answer?
three
(b) Sometimes the proper number of significant figures will be required for your answer, not just three. In this case, you will see the significant figure icon just before a question that checks for the correct number of significant figures. For example, give the number, 143.488, to 2 significant figures.
140
- 1) ALL non-zero numbers (1,2,3,4,5,6,7,8,9) are ALWAYS significant.
- 2) ALL zeroes between non-zero numbers are ALWAYS significant.
- 3) ALL zeroes which are SIMULTANEOUSLY to the right of the decimal point AND at the end of the number are ALWAYS significant.
- 4) ALL zeroes which are to the left of a written decimal point and are in a number >= 10 are ALWAYS significant.
(c) Enter the charge of an electron (1.6 10-19 C) using the scientific "E" notation so that the number can be read by WebAssign.
1.6e-19 C
Just replace the " * 10^ " with a " E "
"times ten to the... " and "E" are the same thing.
You're just bringing the exponent down and taking away the ten, the E separates the portions of the number just like *10^ did
(d) Multiply 3.7 and 0.280 and enter the answer according to WebAssign's 1% tolerance or three-significant-figures rule for numerical answers.
3.7*.280
Stoichiometry
Solutions of sulfuric acid and lead 2 acetate react to form solid lead 2 sulfate and a solution of acetic acid. 7.30g of sulfuric acid and 7.30g of lead 2 acetate are mixed, calculate the number of grams of each of the following present in the mixture after the reaction is complete.
H2SO4 + Pb(CH3COO)2 = PbSO4 + 2 CH3COOH
moles H2SO4 = 7.30 g / 98.078 g/mol= 0.0744
moles Pb(CH3COO)2 = 7.30 g / 325.3 g/mol=0.0224
moles H2SO4 in excess = 0.0744 - 0.0224 = 0.0520
mass H2SO4 in excess = 0.0520 mol x 98.078 g/mol=5.10 g
mass lead acetate = 0
moles acetic acid = 2 x 0.0224 = 0.0448
mass acetic acid = 0.0448 mol x 60.054 g/mol=2.69 g
Give the empirical formula of each of the following compounds if a sample contains the following quantities of each element.
(a) 0.0455 mol C, 0.039 mol H, and 0.013 mol O
divide by the smallest number
0.0455/ 0.013 = 3.5 => C
0.039/ 0.013 = 3 => H
0.013/ 0.013 = 1 => O
multiply by 2 to get whole numbers
C7H6O2
(b) 23.4 g C, 3.9 g H, and 20.8 g O
moles C = 23.4 / 12.011 g/mol= 1.95
moles H = 3.9 / 1.008 g/mol= 3.87
moles O = 20.8 / 15.9994 g/mol= 1.30
1.95/1.30 = 1.5 => C
3.87/ 1.30= 3 => H
1.30/ 1.30 = 1 => O
C3H6O2
(c) 69.9% iron and 30.1% oxygen by mass
moles Fe = 69.9/ 55.847 = 1.25
moles O = 30.1 / 15.9994 = 1.88
1.25/1.25 = 1 => Fe
1.88/ 1.25 = 1.5 => O
Fe2O3
What is the molecular formula of each of the following compounds?
(a) empirical formula HCO2, molar mass = 90.0 g /mol
Mass of HCO2
H – 1
C – 12
O – 16(2) = 32
32 + 12 + 1 = 45
90/45 = 2 Multiply the subscripts of the emperical formula’s elements by a factor of 2.
H2C2O4
(b) empirical formula C2H4O, molar mass = 88 g /mol
C – 12(2)
H – 4(1)
O – 16(1)
24 + 4 + 16 = 44
88/44 = 2 Multiply the subscripts of the emperical formula’s elements by a factor of 2.
C4H8O2
(a) The characteristic odor of pineapple is due to ethyl
butyrate, a compound containing carbon, hydrogen, and oxygen. Combustion of 5.84
mg of ethyl butyrate produces 13.27 mg of CO2 and 5.42 mg of H2O. What is the
empirical formula of the compound?
C3H6O
(b) Nicotine, a component of tobacco, is composed of C, H, and N. A 3.150-mg
sample of nicotine was combusted, producing 8.545 mg of CO2 and 2.450 mg of H2O.
What is the empirical formula for
nicotine?
C5H7N
If nicotine has a molar mass of 160 ± 5 g/mol, what is its molecular formula? -
C – 12(5) =
60
H – 1(7) = 7
N – 14(1) =14
60 + 7 + 14 = 81
81*x =160 ± 5,
the value of 2 for x yields 162 which is in the given range of 160 ±
5
Multiply the subscripts of the formula’s elements by a factor of
2
C5H7N(2) = C10H14N2
H2SO4 + Pb(CH3COO)2 = PbSO4 + 2 CH3COOH
moles H2SO4 = 7.30 g / 98.078 g/mol= 0.0744
moles Pb(CH3COO)2 = 7.30 g / 325.3 g/mol=0.0224
moles H2SO4 in excess = 0.0744 - 0.0224 = 0.0520
mass H2SO4 in excess = 0.0520 mol x 98.078 g/mol=5.10 g
mass lead acetate = 0
moles acetic acid = 2 x 0.0224 = 0.0448
mass acetic acid = 0.0448 mol x 60.054 g/mol=2.69 g
Give the empirical formula of each of the following compounds if a sample contains the following quantities of each element.
(a) 0.0455 mol C, 0.039 mol H, and 0.013 mol O
divide by the smallest number
0.0455/ 0.013 = 3.5 => C
0.039/ 0.013 = 3 => H
0.013/ 0.013 = 1 => O
multiply by 2 to get whole numbers
C7H6O2
(b) 23.4 g C, 3.9 g H, and 20.8 g O
moles C = 23.4 / 12.011 g/mol= 1.95
moles H = 3.9 / 1.008 g/mol= 3.87
moles O = 20.8 / 15.9994 g/mol= 1.30
1.95/1.30 = 1.5 => C
3.87/ 1.30= 3 => H
1.30/ 1.30 = 1 => O
C3H6O2
(c) 69.9% iron and 30.1% oxygen by mass
moles Fe = 69.9/ 55.847 = 1.25
moles O = 30.1 / 15.9994 = 1.88
1.25/1.25 = 1 => Fe
1.88/ 1.25 = 1.5 => O
Fe2O3
What is the molecular formula of each of the following compounds?
(a) empirical formula HCO2, molar mass = 90.0 g /mol
Mass of HCO2
H – 1
C – 12
O – 16(2) = 32
32 + 12 + 1 = 45
90/45 = 2 Multiply the subscripts of the emperical formula’s elements by a factor of 2.
H2C2O4
(b) empirical formula C2H4O, molar mass = 88 g /mol
C – 12(2)
H – 4(1)
O – 16(1)
24 + 4 + 16 = 44
88/44 = 2 Multiply the subscripts of the emperical formula’s elements by a factor of 2.
C4H8O2
(a) The characteristic odor of pineapple is due to ethyl
butyrate, a compound containing carbon, hydrogen, and oxygen. Combustion of 5.84
mg of ethyl butyrate produces 13.27 mg of CO2 and 5.42 mg of H2O. What is the
empirical formula of the compound?
C3H6O
(b) Nicotine, a component of tobacco, is composed of C, H, and N. A 3.150-mg
sample of nicotine was combusted, producing 8.545 mg of CO2 and 2.450 mg of H2O.
What is the empirical formula for
nicotine?
C5H7N
If nicotine has a molar mass of 160 ± 5 g/mol, what is its molecular formula? -
C – 12(5) =
60
H – 1(7) = 7
N – 14(1) =14
60 + 7 + 14 = 81
81*x =160 ± 5,
the value of 2 for x yields 162 which is in the given range of 160 ±
5
Multiply the subscripts of the formula’s elements by a factor of
2
C5H7N(2) = C10H14N2
Empirical Formula Practice
1) The characteristic odor of pineapple is due to ethyl butyrate, a compound containing carbon, hydrogen, and oxygen. Combustion of 5.84 mg of ethyl butyrate produces 13.27 mg of CO2 and 5.42 mg of H2O. What is the empirical formula of the compound?
C3H6O
2(a) Nicotine, a component of tobacco, is composed of C, H, and N. A 3.150-mg sample of nicotine was combusted, producing 8.545 mg of CO2 and 2.450 mg of H2O. What is the empirical formula for nicotine?
C5H7N
2(b) If nicotine has a molar mass of 160 ± 5 g/mol, what is its molecular formula?
C5H6O is 5(12) + 7(1) + 1(14) = 5881*X = 160 ± 5
81(2) = 162; X = 2
Multiply C5H6O's subscripts by a factor of 2
C10H14N2
C3H6O
2(a) Nicotine, a component of tobacco, is composed of C, H, and N. A 3.150-mg sample of nicotine was combusted, producing 8.545 mg of CO2 and 2.450 mg of H2O. What is the empirical formula for nicotine?
C5H7N
2(b) If nicotine has a molar mass of 160 ± 5 g/mol, what is its molecular formula?
C5H6O is 5(12) + 7(1) + 1(14) = 5881*X = 160 ± 5
81(2) = 162; X = 2
Multiply C5H6O's subscripts by a factor of 2
C10H14N2
Many ions and compounds have very similar names, and there is great potential for confusing them. Write the correct chemical formulas to distinguish between.
(a) calcium sulfide and calcium hydrogen sulfide
calcium sulfide: CaS
calcium hydrogen sulfide: Ca(HS)_2
(b) hydrobromic acid and bromic acid
hydrobromic acid: HBr
bromic acid: HBrO_3
(c) aluminum nitride and aluminum nitrite
aluminum nitride: AlN
aluminum nitrite: Al(NO_2)_3
(d) iron(II) oxide and iron(III) oxide
iron(II) oxide: FeO
iron(III) oxide: Fe_2O_3
(e) ammonia and ammonium ion
ammonia: NH_3
ammonium ion: NH_4^+
(f) potassium sulfite and potassium bisulfite
potassium sulfite: K_2SO_3
potassium bisulfite: KHSO_3
(g) mercurous chloride and mercuric chloride
mercurous chloride: Hg_2Cl_2
mercuric chloride: HgCl_2
(h) chloric acid and perchloric acid
chloric acid: HClO_3
perchloric acid: HClO_4
(a) calcium sulfide and calcium hydrogen sulfide
calcium sulfide: CaS
calcium hydrogen sulfide: Ca(HS)_2
(b) hydrobromic acid and bromic acid
hydrobromic acid: HBr
bromic acid: HBrO_3
(c) aluminum nitride and aluminum nitrite
aluminum nitride: AlN
aluminum nitrite: Al(NO_2)_3
(d) iron(II) oxide and iron(III) oxide
iron(II) oxide: FeO
iron(III) oxide: Fe_2O_3
(e) ammonia and ammonium ion
ammonia: NH_3
ammonium ion: NH_4^+
(f) potassium sulfite and potassium bisulfite
potassium sulfite: K_2SO_3
potassium bisulfite: KHSO_3
(g) mercurous chloride and mercuric chloride
mercurous chloride: Hg_2Cl_2
mercuric chloride: HgCl_2
(h) chloric acid and perchloric acid
chloric acid: HClO_3
perchloric acid: HClO_4
Brown & LeMay Ch 1
Classify each of the following as a pure substance or a mixture; if a mixture, indicate whether it is homogeneous or heterogeneous.
(a) seawater
mixture, homogeneous
(b) graphite
pure substance
(c) beer
mixture, heterogeneous
(d) diesel gasoline
mixture, heterogeneous
Give the chemical symbol or chemical name, as appropriate, for each of the following elements.
Look them up on any periodic table of elements. http://www.ptable.com/
A solid white substance A is heated strongly in the absence of air. It decomposes to form a new white substance B and a gas C. The gas has exactly the same properties as the product obtained when carbon is burned in an excess of oxygen.
(a) What is solid A most likely to be?
compound
(b) What is solid B most likely to be?
cannot be determined
(c) What is gas C most likely to be?
compound
Read the following description of the element zinc, and indicate which are physical properties and which are chemical properties.
a) Zinc is a silver-gray colored metal.
physical
b) Zinc melts at 420°C.
physical
c) When zinc granules are added to dilute sulfuric acid, hydrogen is given off and the metal dissolves.
chemical
d) Zinc has a hardness on the Mohs scale of 2.5.
physical
e) Zinc has a density of 7.13 g/cm3 at 25°C.
physical
f) Zinc reacts slowly with oxygen gas at elevated temperatures to form zinc oxide, ZnO.
chemical
A match is lit and held under a cold piece of metal. The following observations are made: the match burns, the metal gets warmer, water condenses on the metal, and soot (carbon) is deposited on the metal. Which of these occurrences are due to physical changes, and which are due to chemical changes?
(a) The match burns.
chemical change
(b) The metal gets warmer.
physical change
(c) Water condenses on the metal.
physical change
(d) Soot (carbon) is deposited on the metal.
physical change
Use appropriate metric prefixes to write the following measurements without use of exponents.
(a) 6.33 10-2 L
1L = 1000mL
6.33 10-2 L = (1000 mL / 1 L) x 6.33 10-2 L = 63.3 mL
(b) 7.7 10-6 s
1µs = 1 x 10-6 s
7.7 10-6 s = 7.7 10-6 s x (1µs / 1 x 10-6 s) = 7.7 µs
(c) 7.0 10-4 m
1m = 1000mm
7.0 10-4 m = 7.0 10-4 m x (1000mm / 1m) = 0.7 mm
(d) 4.10 10-9 m3
1L = 1 x 10-3 m3
1µL = 1 x 10-6 L
4.10 10-9 m3 = 4.10 10-9 m3 x (1L / 1 x 10-3 m3) x (1µL /1 x 10-6 L) = 4.1 µL
(e) 1.39 10-7 kg
1kg = 1000g
1g = 1000mg
1.39 10-7 kg = 1.39 10-7 kg x (1000g / 1kg) x (1000mg / 1g) = 0.139 mg
(f) 4.7 10-10 g
1ng = 1 x 10-9 g
4.7 10-10 g = 4.7 10-10 g x (1ng / 1 x 10-9 g) = 0.47 ng
(g) 6.61 109 fs
1µs = 1 x 10-6 s
1fs = 1 x 10-15 s
6.61 109 fs = 6.61 109 fs x (1 x 10-15 s / 1fs) x (1µs / 1 x 10-6 s) = 6.61 µs
(a) A sample of carbon tetrachloride, a liquid once used in dry cleaning, has a mass of 31.8 g and a volume of 20 mL at 25°C. What is its density at this temperature?
Density = mass/volume
31.8/20 = 1.59 g/cm3
(b) The density of platinum is 21.45 g/cm3 at 20°C. Calculate the mass of 73.0 cm3 of platinum at this temperature.
1570 g
(c) The density of magnesium is 1.738 g/cm3 at 20°C. What is the volume of 267 g of this metal at this temperature?
154 cm3
(a) To identify a liquid substance, a student determined its density. Using a graduated cylinder, she measured out a 55-mL sample of the substance. She then measured the mass of the sample, finding that it weighed 43.2 g. She knew that the substance had to be either isopropyl alcohol (density 0.785 g/mL) or toluene (density 0.866 g/mL). What is the calculated density of the substance?
0.79 g/mL
What is the probable identity of the substance?
isopropyl alcohol
(b) An experiment requires 45.6 g of ethylene glycol, a liquid whose density is 1.114 g/mL. Rather than weigh the sample on a balance, a chemist chooses to dispense the liquid using a graduated cylinder. What volume of the liquid should he use?
40.9 mL
(c) A cubic piece of metal measures 5.00 cm on each edge. If the metal is nickel, whose density is 8.90 g/cm3, what is the mass of the cube?
1110 g
Indicate which of the following are exact or inexact numbers.
(a) the number of grams in a kilogram
exact number
(b) the number of months in a year
exact number
(c) the number of students in your chemistry class
exact number
(d) the number of inches in a foot
exact number
(e) the number of mL in a cubic meter of water
exact number
(f) the surface area of a quarter
inexact number
What is the number of significant figures in each of the following measured quantities?
1) ALL non-zero numbers (1,2,3,4,5,6,7,8,9) are ALWAYS significant.
2) ALL zeroes between non-zero numbers are ALWAYS significant.
3) ALL zeroes which are SIMULTANEOUSLY to the right of the decimal point AND at the end of the number are ALWAYS significant.
4) ALL zeroes which are to the left of a written decimal point and are in a number >= 10 are ALWAYS significant.
(a) 353 kg
3
(b) 0.057 s
2
(c) 6.3060 cm
5
(d) 0.0100 L
3
(e) 7.0700 104 cm
5
Carry out the following operations, and express the answer with the appropriate number of significant figures.
Addition & Subtraction Rules: The final value must have only as many decimals as the original value with the least number of decimal places. Multiplication and Division Rules: The final value can only have as many significant figures as the original value with the least significant figures.
(a) 320.43 − (3104.1/1.4)
-1900
(b) [(2.844 107) − (2.900 103)] 2.8965
8.237e+07
(c) (0.0049 20032.0) + (3590. 24)
86000
(d) 865 [1265 − (3.39 114)]
7.60e+05
Perform the following conversions.
(a) 0.068 L to mL
68 mL
(b) 3.0 10-8 m to nm
30. nm
(c) 6.90 105 ns to s
0.000690 s
(d) 1.30 kg/m3 to g/L
1.30 g/L
A copper refinery produces a copper ingot weight 130. lb. If the copper is drawn into wire whose diameter is 8.45mm how many feet of copper can be obtained from the ingot? The density of copper is 8.94 g/cm3 (Assume that the wire is a cylinder whose volume is V = πr2h, where r is the radius and h is its height or length.)
With the mass and density known, you can now calculate the volume of the copper that you have.
59,020g / 8.94g/mL = 6601.79 mL of copper. Now we know that the wire has a diameter of 0.845 centimeters and a wire is basically a cylinder, so the volume is equal to pi*r*r*h where h would be the length of the wire. We have the value of pi, we have the diameter, and we have the volume so calculating h should be easy.
6601.79 mL = pi*0.4225*0.4225*h.
h = 11772 centimeters.
There are about 2.54 centimeters in an inch. Convert cm to feet = 386.22 ft
Two students determine the percentage of lead in a sample as a laboratory exercise. The true percentage is 19.42%. The students' results for three determinations are as follows:
1. 19.3, 19.4, 19.5
2. 19.31, 19.28, 19.34
(a) Calculate the average percentage for each set of data, and tell which set is the more accurate based on the average.
Calculate average by adding the 3 numbers and dividing their sum by the total number of numbers (3)
data set 1
19.4 %
data set 2
19.31 %
Set 1 is more accurate because 19.4 is closer to 19.42 (the true %) than 19.31 % is.
(b) Precision can be judged by examining the average of the deviations from the average value for that data set. (Calculate the average value for each data set, then calculate the average value of the absolute deviations of each measurement from the average.) Which set is more precise to the hundredth place?
data set 1
0.02
data set 2
0.02
Both sets have the same precision.
What type of quantity (for example, length, volume, density) do the following units indicate?
(a) mL
volume
(b) cm2
area
(c) mm3
volume
(d) mg/L
density
(e) ps
time
(f) nm
length
(g) K
temperature
(a) You are given a bottle that contains 4.59 cm3 of a metallic solid. The total mass of the bottle and solid is 35.66 g. The empty bottle weighs 14.23 g. What is the density of the solid?
Mass of bottle − empty bottle = mass of metallic solid
35.66 g − 14.23 g = 21.43 g
Density = Mass/Volume
x g/cm³ = (21.43 g) / (4.59 cm³) = 4.70 g/cm³
(b) Mercury is traded by the "flask," a unit that has a mass of 34.5 kg. What is the volume of a flask of mercury if the density of mercury is 13.5 g/mL?
Volume = Mass/Density
x mL = (34.5 kg) / (13.5 g/mL)
x mL = (34500 g) / (13.5 g/mL)
= 2555.55 mL = 2.56 L
(c) A thief plans to steal a gold sphere with a radius of 27.7 cm. from a museum. If the gold has a density of 19 g/cm3, what is the mass of the sphere? [The volume of a sphere is V = (4/3)π r3.]
V = 4/3 pi r ^3
Where r = 27.3 cm plug in and do the arithmetic.
V = 4/3*3.14*27.3 cm^3
V = 4/3*3.14*20,300 cm^3 = 85,000 cm^3
85000 cm^3*19.3 g/cm^3 = 1,640,000 g /1000 g = 1,640 Kg
Is he likely to be able to walk off with it unassisted?
No! That's about the weight of a car!
Significant Figures Tutorial
The other rules are extremely easy.
Rule 10: Consider only those values explicitly given in the problem.
Enter the value of R for the specified units (make sure you pick the value with the correct units from among the more than a dozen listed). Specify at least as many significant figures as you calculated for the value of lnK.
8.31 J·K-1·mol-1
In order to solve the equation correctly, we need the value of R in kJ·K-1·mol-1, so we need to divide it by 1000 (exactly). Enter the value of R in kJ·K-1·mol-1 to the same number of significant figures as above.
.00831 kJ·K-1·mol-1
Now, calculate the value of ΔG° if T is 298 K.
-6.08 kJ/mol
(a) seawater
mixture, homogeneous
(b) graphite
pure substance
(c) beer
mixture, heterogeneous
(d) diesel gasoline
mixture, heterogeneous
Give the chemical symbol or chemical name, as appropriate, for each of the following elements.
Look them up on any periodic table of elements. http://www.ptable.com/
A solid white substance A is heated strongly in the absence of air. It decomposes to form a new white substance B and a gas C. The gas has exactly the same properties as the product obtained when carbon is burned in an excess of oxygen.
(a) What is solid A most likely to be?
compound
(b) What is solid B most likely to be?
cannot be determined
(c) What is gas C most likely to be?
compound
Read the following description of the element zinc, and indicate which are physical properties and which are chemical properties.
a) Zinc is a silver-gray colored metal.
physical
b) Zinc melts at 420°C.
physical
c) When zinc granules are added to dilute sulfuric acid, hydrogen is given off and the metal dissolves.
chemical
d) Zinc has a hardness on the Mohs scale of 2.5.
physical
e) Zinc has a density of 7.13 g/cm3 at 25°C.
physical
f) Zinc reacts slowly with oxygen gas at elevated temperatures to form zinc oxide, ZnO.
chemical
A match is lit and held under a cold piece of metal. The following observations are made: the match burns, the metal gets warmer, water condenses on the metal, and soot (carbon) is deposited on the metal. Which of these occurrences are due to physical changes, and which are due to chemical changes?
(a) The match burns.
chemical change
(b) The metal gets warmer.
physical change
(c) Water condenses on the metal.
physical change
(d) Soot (carbon) is deposited on the metal.
physical change
Use appropriate metric prefixes to write the following measurements without use of exponents.
(a) 6.33 10-2 L
1L = 1000mL
6.33 10-2 L = (1000 mL / 1 L) x 6.33 10-2 L = 63.3 mL
(b) 7.7 10-6 s
1µs = 1 x 10-6 s
7.7 10-6 s = 7.7 10-6 s x (1µs / 1 x 10-6 s) = 7.7 µs
(c) 7.0 10-4 m
1m = 1000mm
7.0 10-4 m = 7.0 10-4 m x (1000mm / 1m) = 0.7 mm
(d) 4.10 10-9 m3
1L = 1 x 10-3 m3
1µL = 1 x 10-6 L
4.10 10-9 m3 = 4.10 10-9 m3 x (1L / 1 x 10-3 m3) x (1µL /1 x 10-6 L) = 4.1 µL
(e) 1.39 10-7 kg
1kg = 1000g
1g = 1000mg
1.39 10-7 kg = 1.39 10-7 kg x (1000g / 1kg) x (1000mg / 1g) = 0.139 mg
(f) 4.7 10-10 g
1ng = 1 x 10-9 g
4.7 10-10 g = 4.7 10-10 g x (1ng / 1 x 10-9 g) = 0.47 ng
(g) 6.61 109 fs
1µs = 1 x 10-6 s
1fs = 1 x 10-15 s
6.61 109 fs = 6.61 109 fs x (1 x 10-15 s / 1fs) x (1µs / 1 x 10-6 s) = 6.61 µs
(a) A sample of carbon tetrachloride, a liquid once used in dry cleaning, has a mass of 31.8 g and a volume of 20 mL at 25°C. What is its density at this temperature?
Density = mass/volume
31.8/20 = 1.59 g/cm3
(b) The density of platinum is 21.45 g/cm3 at 20°C. Calculate the mass of 73.0 cm3 of platinum at this temperature.
1570 g
(c) The density of magnesium is 1.738 g/cm3 at 20°C. What is the volume of 267 g of this metal at this temperature?
154 cm3
(a) To identify a liquid substance, a student determined its density. Using a graduated cylinder, she measured out a 55-mL sample of the substance. She then measured the mass of the sample, finding that it weighed 43.2 g. She knew that the substance had to be either isopropyl alcohol (density 0.785 g/mL) or toluene (density 0.866 g/mL). What is the calculated density of the substance?
0.79 g/mL
What is the probable identity of the substance?
isopropyl alcohol
(b) An experiment requires 45.6 g of ethylene glycol, a liquid whose density is 1.114 g/mL. Rather than weigh the sample on a balance, a chemist chooses to dispense the liquid using a graduated cylinder. What volume of the liquid should he use?
40.9 mL
(c) A cubic piece of metal measures 5.00 cm on each edge. If the metal is nickel, whose density is 8.90 g/cm3, what is the mass of the cube?
1110 g
Indicate which of the following are exact or inexact numbers.
(a) the number of grams in a kilogram
exact number
(b) the number of months in a year
exact number
(c) the number of students in your chemistry class
exact number
(d) the number of inches in a foot
exact number
(e) the number of mL in a cubic meter of water
exact number
(f) the surface area of a quarter
inexact number
What is the number of significant figures in each of the following measured quantities?
1) ALL non-zero numbers (1,2,3,4,5,6,7,8,9) are ALWAYS significant.
2) ALL zeroes between non-zero numbers are ALWAYS significant.
3) ALL zeroes which are SIMULTANEOUSLY to the right of the decimal point AND at the end of the number are ALWAYS significant.
4) ALL zeroes which are to the left of a written decimal point and are in a number >= 10 are ALWAYS significant.
(a) 353 kg
3
(b) 0.057 s
2
(c) 6.3060 cm
5
(d) 0.0100 L
3
(e) 7.0700 104 cm
5
Carry out the following operations, and express the answer with the appropriate number of significant figures.
Addition & Subtraction Rules: The final value must have only as many decimals as the original value with the least number of decimal places. Multiplication and Division Rules: The final value can only have as many significant figures as the original value with the least significant figures.
(a) 320.43 − (3104.1/1.4)
-1900
(b) [(2.844 107) − (2.900 103)] 2.8965
8.237e+07
(c) (0.0049 20032.0) + (3590. 24)
86000
(d) 865 [1265 − (3.39 114)]
7.60e+05
Perform the following conversions.
(a) 0.068 L to mL
68 mL
(b) 3.0 10-8 m to nm
30. nm
(c) 6.90 105 ns to s
0.000690 s
(d) 1.30 kg/m3 to g/L
1.30 g/L
A copper refinery produces a copper ingot weight 130. lb. If the copper is drawn into wire whose diameter is 8.45mm how many feet of copper can be obtained from the ingot? The density of copper is 8.94 g/cm3 (Assume that the wire is a cylinder whose volume is V = πr2h, where r is the radius and h is its height or length.)
With the mass and density known, you can now calculate the volume of the copper that you have.
59,020g / 8.94g/mL = 6601.79 mL of copper. Now we know that the wire has a diameter of 0.845 centimeters and a wire is basically a cylinder, so the volume is equal to pi*r*r*h where h would be the length of the wire. We have the value of pi, we have the diameter, and we have the volume so calculating h should be easy.
6601.79 mL = pi*0.4225*0.4225*h.
h = 11772 centimeters.
There are about 2.54 centimeters in an inch. Convert cm to feet = 386.22 ft
Two students determine the percentage of lead in a sample as a laboratory exercise. The true percentage is 19.42%. The students' results for three determinations are as follows:
1. 19.3, 19.4, 19.5
2. 19.31, 19.28, 19.34
(a) Calculate the average percentage for each set of data, and tell which set is the more accurate based on the average.
Calculate average by adding the 3 numbers and dividing their sum by the total number of numbers (3)
data set 1
19.4 %
data set 2
19.31 %
Set 1 is more accurate because 19.4 is closer to 19.42 (the true %) than 19.31 % is.
(b) Precision can be judged by examining the average of the deviations from the average value for that data set. (Calculate the average value for each data set, then calculate the average value of the absolute deviations of each measurement from the average.) Which set is more precise to the hundredth place?
data set 1
0.02
data set 2
0.02
Both sets have the same precision.
What type of quantity (for example, length, volume, density) do the following units indicate?
(a) mL
volume
(b) cm2
area
(c) mm3
volume
(d) mg/L
density
(e) ps
time
(f) nm
length
(g) K
temperature
(a) You are given a bottle that contains 4.59 cm3 of a metallic solid. The total mass of the bottle and solid is 35.66 g. The empty bottle weighs 14.23 g. What is the density of the solid?
Mass of bottle − empty bottle = mass of metallic solid
35.66 g − 14.23 g = 21.43 g
Density = Mass/Volume
x g/cm³ = (21.43 g) / (4.59 cm³) = 4.70 g/cm³
(b) Mercury is traded by the "flask," a unit that has a mass of 34.5 kg. What is the volume of a flask of mercury if the density of mercury is 13.5 g/mL?
Volume = Mass/Density
x mL = (34.5 kg) / (13.5 g/mL)
x mL = (34500 g) / (13.5 g/mL)
= 2555.55 mL = 2.56 L
(c) A thief plans to steal a gold sphere with a radius of 27.7 cm. from a museum. If the gold has a density of 19 g/cm3, what is the mass of the sphere? [The volume of a sphere is V = (4/3)π r3.]
V = 4/3 pi r ^3
Where r = 27.3 cm plug in and do the arithmetic.
V = 4/3*3.14*27.3 cm^3
V = 4/3*3.14*20,300 cm^3 = 85,000 cm^3
85000 cm^3*19.3 g/cm^3 = 1,640,000 g /1000 g = 1,640 Kg
Is he likely to be able to walk off with it unassisted?
No! That's about the weight of a car!
Significant Figures Tutorial
The other rules are extremely easy.
Rule 10: Consider only those values explicitly given in the problem.
Enter the value of R for the specified units (make sure you pick the value with the correct units from among the more than a dozen listed). Specify at least as many significant figures as you calculated for the value of lnK.
8.31 J·K-1·mol-1
In order to solve the equation correctly, we need the value of R in kJ·K-1·mol-1, so we need to divide it by 1000 (exactly). Enter the value of R in kJ·K-1·mol-1 to the same number of significant figures as above.
.00831 kJ·K-1·mol-1
Now, calculate the value of ΔG° if T is 298 K.
-6.08 kJ/mol
Brown and LeMay Ch 2, 25
A chemist finds that 30.82 g of nitrogen will react with 17.60 g, 35.20 g, 70.40 g, or 88.00 g of oxygen to form four different compounds.
(a) Calculate the mass of oxygen per gram of nitrogen in each compound.
first compound
1 x 17.60 / 30.82 = 0.5711 g of O
second compound
1 x 35.20 / 30.82 = 1.142 g of O
third compound
1 x 70.40 / 30.82 = 2.284 g of O
fourth compound
1 x 88.00 / 30.82 = 2.855 g of O
(b) How do the numbers in part (a) support Dalton's atomic theory?
These masses of oxygen per one gram nitrogen are in the ratio of 1:2:4:5 and thus obey the law of multiple proportions.
This condition arises because atoms are the indivisible entities combining, as stated in Dalton's theory. Since atoms are indivisible, they must combine in ratios of small whole numbers.
A negatively charged particle is caused to move between two electrically charged plates, as illustrated in the figure below.
(a) Why does the path of the charged particle bend?
its charge and the charges on the plates
(b) As the charge on the plates is increased, would you expect the bending to increase, decrease, or stay the same?
increase
(c) As the mass of the particle is increased while the speed of the particles remains the same, would you expect the bending to increase, decrease, or stay the same?
decrease
(d) An unknown particle is sent through the apparatus. Its path is deflected in the opposite direction from the negatively charged particle, and it is deflected by a smaller magnitude. What can you conclude about this unknown particle? (Select all that apply.)
The particle has less mass than the electron. The particle is negatively charged.
The radius of an atom of krypton (Kr) is about 1.9 Å.
(a) Express this distance in nanometers (nm).
1 angstroms = 0.1 nanometers
.19 nm
Express this distance in picometers (pm).
1 angstroms = 100 picometers
190 pm
(b) How many krypton atoms would have to be lined up to span 6.5 mm?
The amount would needed to span 1 mm would be easy, just convert .19 nm into mm, which will be .00000019 mm. Divide 6.5 by that number and you will have 3.4 x 10^6 and divide by two, because it would be side to side (diameter) and you will have 1.7x10^6 Krypton atoms.
No of atoms=
1mm/ 1.9 angstrom
= 10^ (-3) /1.9 * 10^(-10)
= 1*[10^7] /1.9
= 0.5263 * 10^7
= 5263157 atoms
(c) If the atom is assumed to be a sphere, what is the volume in cm3 of a krypton (Kr) single atom?
Volume= 4/3 )( r^3
= 4/3 * 3.14 * [1.9*10^(-8)]^3 cm^3
= 28.71634* 10^(-24) cm^3
How many protons, neutrons, and electrons are in the following atoms.
Refer to http://www.ptable.com/
192 Po +3
The 192 is the mass number, the sum of the protons and then
neutrons. The symbol Po gives us the protons, 84.
Neutrons ---> 192
minus 84 = 108
When the atom is neutral, the number of protons and electrons are equal. A +3 ion has lost 3 electrons, to make 81 electrons in 192
Po +3
Copper has two naturally occurring isotopes, 63Cu (isotopic mass 62.9396 amu) and 65Cu (isotopic mass 64.9278 amu). If copper has an atomic mass of 63.546 amu, what is the percent abundance of each copper isotope?
let x = % abundance 63Cu
let y = % abundance 65Cu
x +y = 100
y = 100-x
63.546 = 62.9396 x + 64.9278 ( 100-x) / 100
6354.6 = 62.9396 x + 6492.78 - 64.9278 x
138.18 = 1.9882 x
x = 69.50 %
y = 100 - 69.50 = 30.50
Write the empirical formula corresponding to each of the following molecular formulas.
(a) C2H4
CH2
(b) P4S10
P2S5
(c) C6H4Cl2
C3H2Cl
(d) S4N2
S2N
(e) C2H4O2
CH2O
(f) C6H6
CH
(g)C4H8O2
C2H4O
(h)B3N3H6
BNH2
How many of the indicated atoms are represented by each chemical formula?
(a) carbon atoms in C2H5COOCH3
2+1 = 4
(b) oxygen atoms in Ca(ClO3)2
3*2 = 6
(c) hydrogen atoms in (NH4)2HPO4?
4*2 + 1 = 9
The most common charge associated with silver in its compounds is 1+. Indicate the chemical formulas you would expect for compounds formed between Ag and each of the following.
(a) nitrogen
Ag3N
(b) oxygen
Ag2O
(c) bromine
AgBr
Which of the following are ionic, and which are molecular?
(a) (NH4)3PO4 ionic
(b)PF5 molecular
(c) P4O10 molecular
(d) SCl2 molecular
(e) FeCl3 ionic
(f) NaI ionic
(g) XeO3 molecular
(h) LaP ionic
(i) NH2OH molecular
(j) (NH4)2SO4 ionic
(k) N2O4 molecular
(l) C2H5Br molecular
(m) Zn(OH)2 ionic
Give the chemical formula for each of the following.
(a) chlorite ion
ClO2-
(b) chloride ion
Cl-
(c) chlorate ion
ClO3-
(d) perchlorate ion
ClO4-
(e) hypochlorite ion
ClO-
Name the following ionic compounds. (Type your answer using the format copper(II) chloride for CuCl2.)
(a) Fe(OH)2
iron(II) hydroxide
(b) Li2O
lithium oxide
(c) Al(OH)3
aluminum(III) hydroxide
(d) Fe2(CO3)3
iron(III) carbonate
(e) Ag2SO4
silver sulfate
(f) Hg2Br2
mercury(I) bromide
(g) Ca(NO3)2
calcium nitrate
(h) Cu(NO3)2
copper(II) nitrate
(i) K2CrO4
potassium chromate
(j) KMnO4
potassium permanganate
(k) MgCO3
magnesium carbonate
(l) Mg3P2
magnesium phosphide
Give the chemical formula for each of the following ionic compounds.
(a) potassium chromate
K2CrO4
(b) iron(III) chloride
FeCl3
(c) sodium hypobromite
NaBrO
(d) potassium permanganate
KMnO4
(e) mercury(I) bromide
Hg2Br2
(f) magnesium nitride
Mg3N2
(g) chromium(III) hydroxide
Cr(OH)3
Give the name or chemical formula, as appropriate, for each of the following acids.
http://www.kentchemistry.com/links/naming/acids.htm
(a) HIO3
iodic acid
(b) HBr
hydrobromic acid
(c) H3PO4
phosphoric acid
(d) bromic acid
HBrO3
(e) hypochlorous acid
HClO
(f) sulfurous acid
H2SO3
The oxides of nitrogen are very important components in urban air pollution. Name each of the following compounds.
(a) N2O
dinitrogen monoxide
(b) NO
nitrogen monoxide
(c) NO2
nitrogen dioxide
(d) N2O5
dinitrogen pentoxide
(e) N2O4
dinitrogen tetroxide
From the following list of elements—Ga, Al, Sb, Kr, Cs, Sr, F, H, S—pick the one that best fits each description; use each element only once.
(a) an alkali metal
Cs
(b) an alkaline earth metal
Sr
(c) a noble gas
Kr
(d) a halogen
F
(e) a metalloid
Sb
(f) a nonmetal listed in group IA
H
(g) a metal that forms a 3+ ion
Al
(h) a nonmetal that forms a 2- ion
S
(i) an element that resembles aluminum
Ga
Organic compounds always contain the element
carbon
Match the name with the chemical formula.
nonane C9H20
decane C10H22
heptane C7H16
butane C4H10
octane C8H18
pentane C5H12
methane CH4
propane C3H8
ethane C2H6
hexane C6H14
WCU-HP3-2
Name the following compound:
(a) Calculate the mass of oxygen per gram of nitrogen in each compound.
first compound
1 x 17.60 / 30.82 = 0.5711 g of O
second compound
1 x 35.20 / 30.82 = 1.142 g of O
third compound
1 x 70.40 / 30.82 = 2.284 g of O
fourth compound
1 x 88.00 / 30.82 = 2.855 g of O
(b) How do the numbers in part (a) support Dalton's atomic theory?
These masses of oxygen per one gram nitrogen are in the ratio of 1:2:4:5 and thus obey the law of multiple proportions.
This condition arises because atoms are the indivisible entities combining, as stated in Dalton's theory. Since atoms are indivisible, they must combine in ratios of small whole numbers.
A negatively charged particle is caused to move between two electrically charged plates, as illustrated in the figure below.
(a) Why does the path of the charged particle bend?
its charge and the charges on the plates
(b) As the charge on the plates is increased, would you expect the bending to increase, decrease, or stay the same?
increase
(c) As the mass of the particle is increased while the speed of the particles remains the same, would you expect the bending to increase, decrease, or stay the same?
decrease
(d) An unknown particle is sent through the apparatus. Its path is deflected in the opposite direction from the negatively charged particle, and it is deflected by a smaller magnitude. What can you conclude about this unknown particle? (Select all that apply.)
The particle has less mass than the electron. The particle is negatively charged.
The radius of an atom of krypton (Kr) is about 1.9 Å.
(a) Express this distance in nanometers (nm).
1 angstroms = 0.1 nanometers
.19 nm
Express this distance in picometers (pm).
1 angstroms = 100 picometers
190 pm
(b) How many krypton atoms would have to be lined up to span 6.5 mm?
The amount would needed to span 1 mm would be easy, just convert .19 nm into mm, which will be .00000019 mm. Divide 6.5 by that number and you will have 3.4 x 10^6 and divide by two, because it would be side to side (diameter) and you will have 1.7x10^6 Krypton atoms.
No of atoms=
1mm/ 1.9 angstrom
= 10^ (-3) /1.9 * 10^(-10)
= 1*[10^7] /1.9
= 0.5263 * 10^7
= 5263157 atoms
(c) If the atom is assumed to be a sphere, what is the volume in cm3 of a krypton (Kr) single atom?
Volume= 4/3 )( r^3
= 4/3 * 3.14 * [1.9*10^(-8)]^3 cm^3
= 28.71634* 10^(-24) cm^3
How many protons, neutrons, and electrons are in the following atoms.
Refer to http://www.ptable.com/
192 Po +3
The 192 is the mass number, the sum of the protons and then
neutrons. The symbol Po gives us the protons, 84.
Neutrons ---> 192
minus 84 = 108
When the atom is neutral, the number of protons and electrons are equal. A +3 ion has lost 3 electrons, to make 81 electrons in 192
Po +3
Copper has two naturally occurring isotopes, 63Cu (isotopic mass 62.9396 amu) and 65Cu (isotopic mass 64.9278 amu). If copper has an atomic mass of 63.546 amu, what is the percent abundance of each copper isotope?
let x = % abundance 63Cu
let y = % abundance 65Cu
x +y = 100
y = 100-x
63.546 = 62.9396 x + 64.9278 ( 100-x) / 100
6354.6 = 62.9396 x + 6492.78 - 64.9278 x
138.18 = 1.9882 x
x = 69.50 %
y = 100 - 69.50 = 30.50
Write the empirical formula corresponding to each of the following molecular formulas.
(a) C2H4
CH2
(b) P4S10
P2S5
(c) C6H4Cl2
C3H2Cl
(d) S4N2
S2N
(e) C2H4O2
CH2O
(f) C6H6
CH
(g)C4H8O2
C2H4O
(h)B3N3H6
BNH2
How many of the indicated atoms are represented by each chemical formula?
(a) carbon atoms in C2H5COOCH3
2+1 = 4
(b) oxygen atoms in Ca(ClO3)2
3*2 = 6
(c) hydrogen atoms in (NH4)2HPO4?
4*2 + 1 = 9
The most common charge associated with silver in its compounds is 1+. Indicate the chemical formulas you would expect for compounds formed between Ag and each of the following.
(a) nitrogen
Ag3N
(b) oxygen
Ag2O
(c) bromine
AgBr
Which of the following are ionic, and which are molecular?
(a) (NH4)3PO4 ionic
(b)PF5 molecular
(c) P4O10 molecular
(d) SCl2 molecular
(e) FeCl3 ionic
(f) NaI ionic
(g) XeO3 molecular
(h) LaP ionic
(i) NH2OH molecular
(j) (NH4)2SO4 ionic
(k) N2O4 molecular
(l) C2H5Br molecular
(m) Zn(OH)2 ionic
Give the chemical formula for each of the following.
(a) chlorite ion
ClO2-
(b) chloride ion
Cl-
(c) chlorate ion
ClO3-
(d) perchlorate ion
ClO4-
(e) hypochlorite ion
ClO-
Name the following ionic compounds. (Type your answer using the format copper(II) chloride for CuCl2.)
(a) Fe(OH)2
iron(II) hydroxide
(b) Li2O
lithium oxide
(c) Al(OH)3
aluminum(III) hydroxide
(d) Fe2(CO3)3
iron(III) carbonate
(e) Ag2SO4
silver sulfate
(f) Hg2Br2
mercury(I) bromide
(g) Ca(NO3)2
calcium nitrate
(h) Cu(NO3)2
copper(II) nitrate
(i) K2CrO4
potassium chromate
(j) KMnO4
potassium permanganate
(k) MgCO3
magnesium carbonate
(l) Mg3P2
magnesium phosphide
Give the chemical formula for each of the following ionic compounds.
(a) potassium chromate
K2CrO4
(b) iron(III) chloride
FeCl3
(c) sodium hypobromite
NaBrO
(d) potassium permanganate
KMnO4
(e) mercury(I) bromide
Hg2Br2
(f) magnesium nitride
Mg3N2
(g) chromium(III) hydroxide
Cr(OH)3
Give the name or chemical formula, as appropriate, for each of the following acids.
http://www.kentchemistry.com/links/naming/acids.htm
(a) HIO3
iodic acid
(b) HBr
hydrobromic acid
(c) H3PO4
phosphoric acid
(d) bromic acid
HBrO3
(e) hypochlorous acid
HClO
(f) sulfurous acid
H2SO3
The oxides of nitrogen are very important components in urban air pollution. Name each of the following compounds.
(a) N2O
dinitrogen monoxide
(b) NO
nitrogen monoxide
(c) NO2
nitrogen dioxide
(d) N2O5
dinitrogen pentoxide
(e) N2O4
dinitrogen tetroxide
From the following list of elements—Ga, Al, Sb, Kr, Cs, Sr, F, H, S—pick the one that best fits each description; use each element only once.
(a) an alkali metal
Cs
(b) an alkaline earth metal
Sr
(c) a noble gas
Kr
(d) a halogen
F
(e) a metalloid
Sb
(f) a nonmetal listed in group IA
H
(g) a metal that forms a 3+ ion
Al
(h) a nonmetal that forms a 2- ion
S
(i) an element that resembles aluminum
Ga
Organic compounds always contain the element
carbon
Match the name with the chemical formula.
nonane C9H20
decane C10H22
heptane C7H16
butane C4H10
octane C8H18
pentane C5H12
methane CH4
propane C3H8
ethane C2H6
hexane C6H14
WCU-HP3-2
Name the following compound:
4-ethyl-2-methylhexane
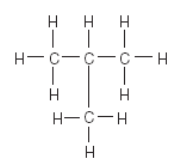
Which formula is an isomer of butane?
but means 4. ane means single bonded
Which formula is an isomer of butane?
but means 4. ane means single bonded
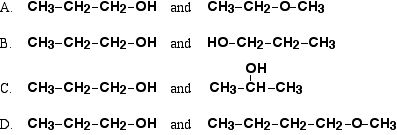
Which pair of compounds are alcohols?
alcohol R-OH
alkene CnH2n
carboxylic acid R-CO2H
amine R-NH2
alkyne CnH2n-2
amide R-CONH
alkane CnH2n+2
alkyl CnH2n+1
Match the following pairs of compounds with the descriptions below:
alkene CnH2n
carboxylic acid R-CO2H
amine R-NH2
alkyne CnH2n-2
amide R-CONH
alkane CnH2n+2
alkyl CnH2n+1
Match the following pairs of compounds with the descriptions below:
Structural isomers with identical functional groups C
Structural isomers with different functional groups A
Not structural isomers, not same functional group D
Identical molecules B
Identify the functional group(s) in each of the following compounds. (Select all that apply.)
(a) CH3CCH2COOH - alkyne, carboxylic acid
(b) ester
(c) alkene, amine
(d) alcohol, halocarbon
(e) alkene, aldehyde
(f) H3C-CH2-OH - alcohol
Write names for the following coordination compounds. (Type your answer using the format copper(II) chloride for CuCl2.)
(a) [Cd(en)Cl2] dichloroethylenediaminecadmium(II)
(b) [Cr(H2O)4Cl2]Cl tetraaquadichlorochromium(III) chloride
(c) [Cr(NH3)5CO3]Cl pentaamminecarbonatochromium(III) chloride
(d) K4[Mn(CN)6] potassium hexacyanomanganate(II)
Structural isomers with different functional groups A
Not structural isomers, not same functional group D
Identical molecules B
Identify the functional group(s) in each of the following compounds. (Select all that apply.)
(a) CH3CCH2COOH - alkyne, carboxylic acid
(b) ester
(c) alkene, amine
(d) alcohol, halocarbon
(e) alkene, aldehyde
(f) H3C-CH2-OH - alcohol
Write names for the following coordination compounds. (Type your answer using the format copper(II) chloride for CuCl2.)
(a) [Cd(en)Cl2] dichloroethylenediaminecadmium(II)
(b) [Cr(H2O)4Cl2]Cl tetraaquadichlorochromium(III) chloride
(c) [Cr(NH3)5CO3]Cl pentaamminecarbonatochromium(III) chloride
(d) K4[Mn(CN)6] potassium hexacyanomanganate(II)
Molarity & Titration
1a) Calculate the molarity of a solution made by dissolving 0.0715 mol Na2SO4 in
enough water to form 650 mL of solution.
molarity (M) is mol/L, so molarity of 0.0715 g Na2SO4 in 600 mL solution is:
Na2's mass + SO4's mass = 142.1 g
(.0715 g)(1 mol/142.1 g) = 5.03e-4 mol, 650 mL = .65 L
2.99e-4 mol/.6 L = 7.74e-4M
1b) How many moles of KMnO4
are present in 25.0 mL of a 0.0850 M solution?
this is like (a) but in reverse. start with 25.0 mL and convert to L (1L/1000
mL) then multiply by the molarity (0.0850 mol/L). this way "L" cancels out and
you are left with your answer in mol.
25/1000 = 0.025
0.025*.0850 = 0.002125 mol
1c) How many
milliliters of 10.6 M HCl solution are needed to obtain 0.305 mol of HCl?
Molarity = moles/volume
10.6 M = 0.305 moles/volume
volume*10.6 = 0.305
volume = 0.305/10.6 = 0.0288 Liters
Since you want the answer in milliliters, multiply 0.0288 by 1,000 and you get 28.8 mL
2) A person suffering from hyponatremia has a sodium ion concentration in the blood of 0.120 M and a total blood volume of 4.9 L.What mass of sodium chloride would need to be added to the blood to bring the sodium ion concentration up to 0.139 M, assuming no change in blood volume?
0.120 M x 4.9 L = 0.588 mol
0.139 M x 4.9 L = 0.6811 mol
0.6811 - 0.588 = 0.0931 mol
Na's mass + Cl's mass = 58.45 g
0.0931 mol x 58.45 g/mol = 5.44 g NaCl
In each of the following pairs, indicate which has the higher concentration of Cl- ion:?
a) 0.10 M CaCl2 or 0.15 M KCl solution,
0.10 M CaCl2 releases 0.20 Molar Cl-
0.15 M KCl solution releases 0.15 molar Cl-
CaCl2
b) 100ml of 0.10 M KCl solution or 400 mL of 0.080M LiCl solution
0.100 M KCL releases 0.1 M
Cl- (0.010 moles Cl- in 100ml)
0.080M LiCl releases 0.08 M Cl- (0.032 moles Cl- in 100ml)
KCl releases the greater Molarity of Cl- though LiCl released more moles of Cl-
c) 0.050 M HCL solution or 0.020 M CdCl2 solution.
0.050 M HCl releases 0.050 Molar Cl- 0.020 M CdCl2 releases 0.040 Molar Cl-
HCl releases the greater concentration of Cl-
(a) How many milliliters of a stock solution of 11.0 M HNO3 would you have to use to prepare 0.450 L of 0.500 M HNO3?
M1V1 = M2V2
11M(V1) = .5M(.45L)
V1 = .0205 L = 20.5 mL
b) If you dilute 21.0 mL of the stock solution to a final volume of 0.500 L, what will be the concentration of the diluted solution?
M1V1 = M2V2
11M(21mL which is the same as 0.021 L) = M2(.5L)
M2 = V1M1 / V2
= 0.021 L x 11.0 M / 0.500 L
= 0.462 M
What volume of 0.115M HClO4 solution is needed to neutralize 50mL of 0.0875M NaOH?
Let HClO4 solution becalled HS. Let NaOH solution be called NS.
50mLNS*.0875 molNaOH/1000 mLNS x 1molHClO4/1 mol NaOH x 1000 mLHS/0.115 mol HClO4 = 38 mL HClO4 solution. Cancel the 1000's before entering numbers into the calculator.
What volume of 0.128 M HCl is needed to neutralize 2.82 g of Mg(OH)2?
Write a balanced equation: Mg(OH)2 + 2HCl → MgCl2 + 2H2O
So 1 mole Mg(OH)2 is neutralized by 2 moles HCl
Thus moles HCl needed = 2 x moles Mg(OH)2
moles = mass / molar mass
moles Mg(OH)2 = 2.82 g / 58.3 g/mol = 0.04837 mol
moles HCl needed = 2 x 0.04837 mol = 0.09674 mol HCl
Molarity = moles / Liters
Therefore Liters = moles / molarity
= 0.09674 mol / 0.128 M = 0.7558 L = 756 ml
If 24.9 mL of AgNO3 is needed to precipitate all the Cl‾ ions in a 785-mg sample of KCl (forming AgCl), what is the molarity of the AgNO3 solution?
Write a balanced equation: AgNO3(aq) + KCl(aq) → AgCl(s) + KNO3(aq)
1 mole AgNO3 reacts with 1 mole KCl
moles KCl = mass / molar mass
= 0.785 g / 74.55 g/mol = 0.01053 mol
Since they react in a 1:1 ratio, moles AgNO3 = moles KCl = 0.01053 mol
Molarity = moles / Liters = 0.01053 mol / 0.0249 L = 0.423 M
If 45.0 mL of 0.114 M HCl solution is needed to neutralize a solution of KOH, how many grams of KOH must be present in the solution?
Write a balanced equation: KOH + HCl → KCl + H2O
1mol KOH reacts with 1 mol HCl
Mol HCl in 45.0ml 0.114M solution: = 45/1000*0.114 = 0.00513mol
Therefore the solution of KOH contained 0.00513mol KOH
Molar mass KOH = 39.1+16+1 = 56.1g/mol
0.00513 mol KOH = 56.1*0.00513 = 0.288g KOH
The distinctive odor of vinegar is due to acetic acid, CH3COOH, which reacts
with sodium hydroxide in the following fashion:
CH3COOH (aq) + NaOH (aq) --> H2O}(l) + NaC2H3O2 (aq)
>>If 3.70 mL of vinegar needs 41.0 mL of 0.150 M NaOH to reach the equivalence point in a titration, how many grams of
acetic acid are in a 2.00 qt sample of this vinegar?
(41.0 mL) x (0.150 M) x (1/1) / (3.70 mL) = 1.662 M acetic acid
(2.00 qt) x (0.94635 L/qt) x (1.662 mol/L) x (60.0522 g CH3COOH/mol) = 189 g CH3COOH
enough water to form 650 mL of solution.
molarity (M) is mol/L, so molarity of 0.0715 g Na2SO4 in 600 mL solution is:
Na2's mass + SO4's mass = 142.1 g
(.0715 g)(1 mol/142.1 g) = 5.03e-4 mol, 650 mL = .65 L
2.99e-4 mol/.6 L = 7.74e-4M
1b) How many moles of KMnO4
are present in 25.0 mL of a 0.0850 M solution?
this is like (a) but in reverse. start with 25.0 mL and convert to L (1L/1000
mL) then multiply by the molarity (0.0850 mol/L). this way "L" cancels out and
you are left with your answer in mol.
25/1000 = 0.025
0.025*.0850 = 0.002125 mol
1c) How many
milliliters of 10.6 M HCl solution are needed to obtain 0.305 mol of HCl?
Molarity = moles/volume
10.6 M = 0.305 moles/volume
volume*10.6 = 0.305
volume = 0.305/10.6 = 0.0288 Liters
Since you want the answer in milliliters, multiply 0.0288 by 1,000 and you get 28.8 mL
2) A person suffering from hyponatremia has a sodium ion concentration in the blood of 0.120 M and a total blood volume of 4.9 L.What mass of sodium chloride would need to be added to the blood to bring the sodium ion concentration up to 0.139 M, assuming no change in blood volume?
0.120 M x 4.9 L = 0.588 mol
0.139 M x 4.9 L = 0.6811 mol
0.6811 - 0.588 = 0.0931 mol
Na's mass + Cl's mass = 58.45 g
0.0931 mol x 58.45 g/mol = 5.44 g NaCl
In each of the following pairs, indicate which has the higher concentration of Cl- ion:?
a) 0.10 M CaCl2 or 0.15 M KCl solution,
0.10 M CaCl2 releases 0.20 Molar Cl-
0.15 M KCl solution releases 0.15 molar Cl-
CaCl2
b) 100ml of 0.10 M KCl solution or 400 mL of 0.080M LiCl solution
0.100 M KCL releases 0.1 M
Cl- (0.010 moles Cl- in 100ml)
0.080M LiCl releases 0.08 M Cl- (0.032 moles Cl- in 100ml)
KCl releases the greater Molarity of Cl- though LiCl released more moles of Cl-
c) 0.050 M HCL solution or 0.020 M CdCl2 solution.
0.050 M HCl releases 0.050 Molar Cl- 0.020 M CdCl2 releases 0.040 Molar Cl-
HCl releases the greater concentration of Cl-
(a) How many milliliters of a stock solution of 11.0 M HNO3 would you have to use to prepare 0.450 L of 0.500 M HNO3?
M1V1 = M2V2
11M(V1) = .5M(.45L)
V1 = .0205 L = 20.5 mL
b) If you dilute 21.0 mL of the stock solution to a final volume of 0.500 L, what will be the concentration of the diluted solution?
M1V1 = M2V2
11M(21mL which is the same as 0.021 L) = M2(.5L)
M2 = V1M1 / V2
= 0.021 L x 11.0 M / 0.500 L
= 0.462 M
What volume of 0.115M HClO4 solution is needed to neutralize 50mL of 0.0875M NaOH?
Let HClO4 solution becalled HS. Let NaOH solution be called NS.
50mLNS*.0875 molNaOH/1000 mLNS x 1molHClO4/1 mol NaOH x 1000 mLHS/0.115 mol HClO4 = 38 mL HClO4 solution. Cancel the 1000's before entering numbers into the calculator.
What volume of 0.128 M HCl is needed to neutralize 2.82 g of Mg(OH)2?
Write a balanced equation: Mg(OH)2 + 2HCl → MgCl2 + 2H2O
So 1 mole Mg(OH)2 is neutralized by 2 moles HCl
Thus moles HCl needed = 2 x moles Mg(OH)2
moles = mass / molar mass
moles Mg(OH)2 = 2.82 g / 58.3 g/mol = 0.04837 mol
moles HCl needed = 2 x 0.04837 mol = 0.09674 mol HCl
Molarity = moles / Liters
Therefore Liters = moles / molarity
= 0.09674 mol / 0.128 M = 0.7558 L = 756 ml
If 24.9 mL of AgNO3 is needed to precipitate all the Cl‾ ions in a 785-mg sample of KCl (forming AgCl), what is the molarity of the AgNO3 solution?
Write a balanced equation: AgNO3(aq) + KCl(aq) → AgCl(s) + KNO3(aq)
1 mole AgNO3 reacts with 1 mole KCl
moles KCl = mass / molar mass
= 0.785 g / 74.55 g/mol = 0.01053 mol
Since they react in a 1:1 ratio, moles AgNO3 = moles KCl = 0.01053 mol
Molarity = moles / Liters = 0.01053 mol / 0.0249 L = 0.423 M
If 45.0 mL of 0.114 M HCl solution is needed to neutralize a solution of KOH, how many grams of KOH must be present in the solution?
Write a balanced equation: KOH + HCl → KCl + H2O
1mol KOH reacts with 1 mol HCl
Mol HCl in 45.0ml 0.114M solution: = 45/1000*0.114 = 0.00513mol
Therefore the solution of KOH contained 0.00513mol KOH
Molar mass KOH = 39.1+16+1 = 56.1g/mol
0.00513 mol KOH = 56.1*0.00513 = 0.288g KOH
The distinctive odor of vinegar is due to acetic acid, CH3COOH, which reacts
with sodium hydroxide in the following fashion:
CH3COOH (aq) + NaOH (aq) --> H2O}(l) + NaC2H3O2 (aq)
>>If 3.70 mL of vinegar needs 41.0 mL of 0.150 M NaOH to reach the equivalence point in a titration, how many grams of
acetic acid are in a 2.00 qt sample of this vinegar?
(41.0 mL) x (0.150 M) x (1/1) / (3.70 mL) = 1.662 M acetic acid
(2.00 qt) x (0.94635 L/qt) x (1.662 mol/L) x (60.0522 g CH3COOH/mol) = 189 g CH3COOH
Molarity, Titrations, and Reactions 2
1a) Calculate the molarity of a solution that contains 0.0345 mol NH4Cl in exactly 415 mL of solution.
M= mol/L
415mL = .415L
M= .0345/.415L = 0.08313
1b) How many moles of HNO3 are present in 35.2 mL of a 2.20 M solution of nitric acid?
M= mol/L
35.2 mL = .0352 L
2.20=mol/.352L
2.20(.352) = 0.7744 mol
1c) How many milliliters of 1.50 M KOH solution are needed to provide 0.110 mol of KOH?
M= mol/L
L = M(mol)
.110 mol KOH / 1.5 M KOH = .073 L
.073 L x 1000 = 73 mL
2a) Calculate the number of grams of solute in 0.250 L of 0.150 M KBr
M= mol/L
A 0.15 M solution contains 0.15 moles in a liter of solution. .250 L of that solution contains 0.15/4 moles = 0.0375
moles since there are 4 0.25L portions in one liter. The molar mass of KBr is (80 + 39) = 119 g/mole. 0.0375 mole, then, is 0.0375 x 119 g/mole = 4.4625 g
2b) Calculate the molar concentration of a solution containing 4.83 g of Ca(NO3)2 in 0.200 L
M= mol/L
1) first find the # moles of the Ca(NO3)2
4.83 g x 1 mol/164.1 g = 0.0294 mol
2) M=0.0294 mol/0.200 L = 0.147 M
2c) Calculate the volume of 1.55 M Na3PO4 in milliliters that contains 5.00 g of solute
M= mol/L
moles = 5.00/ 164 =0.0305
V = 0.0305/ 1.57=0.0194 L = 19.4 mL
3a) How many milliliters of 0.155 M HCl are needed to completely neutralize 35.0 mL of 0.101 M Ba(OH)2 solution?
Equation reveals that you need 2 units of HCl to neutralize 1 unit Ba(OH)2
2HCl(aq) + Ba(OH)2(aq) → 2H2O + BaCl2 (aq)
35mL of 0.101M Barium Hydroxide contains 0.035 x 0.101 moles = 0.003535
moles therefore 2 x 0.003535 moles HCl needed = 0.00707 moles HCl to
get the volume, we know the HCl is 0.155M and there is a total of 0.00707 moles needed, so
M= mol/L
re-arrange equation for...
L HCl = 0.00707 / 0.155 = 0.0456 L = 45.6mL
3b) How many milliliters of 3.50 M H2SO4 are needed to neutralize 75.0 g of NaOH?
2NaOH + H2SO4 -> 2H2O + Na2SO4
This tells us the ratio of NaOH : H2SO4 is 2:1
Convert NaOH to mole:
75 g of NaOH = 75 / (23 + 16 + 1) = 1.875 mol
This means that only half this amount of H2SO4 is required to react because of the 2:1 ratio.
So n(H2SO4) = 0.9375 mol.
Since the concentration is 3.50 M, we can use c = n/v rearranged to v = n/c.
v = 0.9375 / 3.50 = 0.267 L = 267 mL
3c) If 56.8 mL of BaCl2 solution is needed to precipitate all the sulfate ion in a 534 mg sample of Na2SO4 (forming BaSO4), what is the molarity of the solution?
Step 1- Convert mg to g, then g to mols of Na2SO4
534mg(1g/1000mg)(1mol/142g) = 0.00376 mols Na2SO4
Step 2 - finding mols SO4 in solution
1 mol SO4 in 1 mol Na2SO4, so there must be 0.00376 mols SO4 in 0.00376mols Na2SO4
Step 3 - finding mols BaCl2
Need 1 mol Ba for every 1 mol SO4 because 1:1 mol ratio between Ba and SO4 in final product BaSO4 so if there are 0.00376mols SO4 there needs to be 0.00376 mols Ba to react with all of the SO4 ions in solution
Step 4 - Calculating molarity of the BaCl2 solution
Molarity = mols/L
1L = 1000mL
Molarity = 0.00376mols BaCl2 / (56.8mL/(1000mL/1L))
Molarity = 0.0662M BaCl2
3d) If 27.5 mL of 0.250 M HCl solution is needed to neutralize a solution of Ca(OH)2, how many grams of Ca(OH)2 must be in the solution?
27.5ml= .0275 liters
.25M *.0275 liters= .006875 mol HCl
.006875mol (due to mole ratio) is halved= .00344mol
.0344mol *74.096(g/mol)= .2547 g
4a) Write the balanced chemical equation for the reaction that occurs. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer.)
NiSO_4(aq) + 2KOH(aq) --> Ni(OH)_2(s) + K_2SO_4(aq)
4b) What precipitate forms? (Omit states-of-matter in your answer.)
Ni(OH)_2
4c) What is the limiting reactant?
KOH
4d) How many grams of this precipitate form?
0.01Mol *(molecular weight of Ni(OH)2)
.01*92.708 = 0.92708 g
4e) What is the concentration of each ion that remains in solution?
Ni++: 0.02Mol/0.3L = .0667 M
K+: 0.02Mol/0.3L = .0667 M
SO4--: 0.1M
5a) The accompanying photo shows the reaction between a solution of Cd(NO3)2 and one of Na2S. What is the identity of the precipitate? (Omit states-of-matter in your answer.)
The reaction is Cd(NO3)2 + Na2S ---> 2NaNO3 + CdS
CdS is the precipitate because NaNO3 is soluble in water.
5b) What ions remain in solution? (Separate substances with a comma.)
Na^+, NO_3^- are the spectator ions
5c)Write the net ionic equation for the reaction. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer.)
Cd^2+(aq) + S^2-(aq) --> CdS(s)
6) Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer.)
(a) Pb(NO3)2(aq) + Na2SO4(aq) → ?
net ionic equation
Pb^2+(aq) + SO_4^2-(aq) --> PbSO_4(s)
spectator ions (Separate substances in a list with a ",". Omit states-of-matter in your answer.)
NO_3^-, Na^+
b) CuBr2(aq) + NaOH(aq) → ?
net ionic equation
Cu^2+(aq) + 2OH^-(aq) --> Cu(OH)_2(s)
spectator ions
Br^-, Na^+
c) AgNO3(aq) + KI(aq) → ?
net ionic equation
Ag^+(aq) + I^-(aq) --> AgI(s)
spectator ions
NO_3^-, K^+
7) Separate samples of a solution of an unknown salt are treated with dilute solutions of HBr, H2SO4, and NaOH. A precipitate forms in all three cases. Which of the following cations could the solution contain?
Both lead(II) and barium ions combine with sulfate ion and hydroxide ion to make insoluble products. But only lead (II) bromide is insoluble, while all K compounds are soluble in water.
The cation is Pb2+
M= mol/L
415mL = .415L
M= .0345/.415L = 0.08313
1b) How many moles of HNO3 are present in 35.2 mL of a 2.20 M solution of nitric acid?
M= mol/L
35.2 mL = .0352 L
2.20=mol/.352L
2.20(.352) = 0.7744 mol
1c) How many milliliters of 1.50 M KOH solution are needed to provide 0.110 mol of KOH?
M= mol/L
L = M(mol)
.110 mol KOH / 1.5 M KOH = .073 L
.073 L x 1000 = 73 mL
2a) Calculate the number of grams of solute in 0.250 L of 0.150 M KBr
M= mol/L
A 0.15 M solution contains 0.15 moles in a liter of solution. .250 L of that solution contains 0.15/4 moles = 0.0375
moles since there are 4 0.25L portions in one liter. The molar mass of KBr is (80 + 39) = 119 g/mole. 0.0375 mole, then, is 0.0375 x 119 g/mole = 4.4625 g
2b) Calculate the molar concentration of a solution containing 4.83 g of Ca(NO3)2 in 0.200 L
M= mol/L
1) first find the # moles of the Ca(NO3)2
4.83 g x 1 mol/164.1 g = 0.0294 mol
2) M=0.0294 mol/0.200 L = 0.147 M
2c) Calculate the volume of 1.55 M Na3PO4 in milliliters that contains 5.00 g of solute
M= mol/L
moles = 5.00/ 164 =0.0305
V = 0.0305/ 1.57=0.0194 L = 19.4 mL
3a) How many milliliters of 0.155 M HCl are needed to completely neutralize 35.0 mL of 0.101 M Ba(OH)2 solution?
Equation reveals that you need 2 units of HCl to neutralize 1 unit Ba(OH)2
2HCl(aq) + Ba(OH)2(aq) → 2H2O + BaCl2 (aq)
35mL of 0.101M Barium Hydroxide contains 0.035 x 0.101 moles = 0.003535
moles therefore 2 x 0.003535 moles HCl needed = 0.00707 moles HCl to
get the volume, we know the HCl is 0.155M and there is a total of 0.00707 moles needed, so
M= mol/L
re-arrange equation for...
L HCl = 0.00707 / 0.155 = 0.0456 L = 45.6mL
3b) How many milliliters of 3.50 M H2SO4 are needed to neutralize 75.0 g of NaOH?
2NaOH + H2SO4 -> 2H2O + Na2SO4
This tells us the ratio of NaOH : H2SO4 is 2:1
Convert NaOH to mole:
75 g of NaOH = 75 / (23 + 16 + 1) = 1.875 mol
This means that only half this amount of H2SO4 is required to react because of the 2:1 ratio.
So n(H2SO4) = 0.9375 mol.
Since the concentration is 3.50 M, we can use c = n/v rearranged to v = n/c.
v = 0.9375 / 3.50 = 0.267 L = 267 mL
3c) If 56.8 mL of BaCl2 solution is needed to precipitate all the sulfate ion in a 534 mg sample of Na2SO4 (forming BaSO4), what is the molarity of the solution?
Step 1- Convert mg to g, then g to mols of Na2SO4
534mg(1g/1000mg)(1mol/142g) = 0.00376 mols Na2SO4
Step 2 - finding mols SO4 in solution
1 mol SO4 in 1 mol Na2SO4, so there must be 0.00376 mols SO4 in 0.00376mols Na2SO4
Step 3 - finding mols BaCl2
Need 1 mol Ba for every 1 mol SO4 because 1:1 mol ratio between Ba and SO4 in final product BaSO4 so if there are 0.00376mols SO4 there needs to be 0.00376 mols Ba to react with all of the SO4 ions in solution
Step 4 - Calculating molarity of the BaCl2 solution
Molarity = mols/L
1L = 1000mL
Molarity = 0.00376mols BaCl2 / (56.8mL/(1000mL/1L))
Molarity = 0.0662M BaCl2
3d) If 27.5 mL of 0.250 M HCl solution is needed to neutralize a solution of Ca(OH)2, how many grams of Ca(OH)2 must be in the solution?
27.5ml= .0275 liters
.25M *.0275 liters= .006875 mol HCl
.006875mol (due to mole ratio) is halved= .00344mol
.0344mol *74.096(g/mol)= .2547 g
4a) Write the balanced chemical equation for the reaction that occurs. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer.)
NiSO_4(aq) + 2KOH(aq) --> Ni(OH)_2(s) + K_2SO_4(aq)
4b) What precipitate forms? (Omit states-of-matter in your answer.)
Ni(OH)_2
4c) What is the limiting reactant?
KOH
4d) How many grams of this precipitate form?
0.01Mol *(molecular weight of Ni(OH)2)
.01*92.708 = 0.92708 g
4e) What is the concentration of each ion that remains in solution?
Ni++: 0.02Mol/0.3L = .0667 M
K+: 0.02Mol/0.3L = .0667 M
SO4--: 0.1M
5a) The accompanying photo shows the reaction between a solution of Cd(NO3)2 and one of Na2S. What is the identity of the precipitate? (Omit states-of-matter in your answer.)
The reaction is Cd(NO3)2 + Na2S ---> 2NaNO3 + CdS
CdS is the precipitate because NaNO3 is soluble in water.
5b) What ions remain in solution? (Separate substances with a comma.)
Na^+, NO_3^- are the spectator ions
5c)Write the net ionic equation for the reaction. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer.)
Cd^2+(aq) + S^2-(aq) --> CdS(s)
6) Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer.)
(a) Pb(NO3)2(aq) + Na2SO4(aq) → ?
net ionic equation
Pb^2+(aq) + SO_4^2-(aq) --> PbSO_4(s)
spectator ions (Separate substances in a list with a ",". Omit states-of-matter in your answer.)
NO_3^-, Na^+
b) CuBr2(aq) + NaOH(aq) → ?
net ionic equation
Cu^2+(aq) + 2OH^-(aq) --> Cu(OH)_2(s)
spectator ions
Br^-, Na^+
c) AgNO3(aq) + KI(aq) → ?
net ionic equation
Ag^+(aq) + I^-(aq) --> AgI(s)
spectator ions
NO_3^-, K^+
7) Separate samples of a solution of an unknown salt are treated with dilute solutions of HBr, H2SO4, and NaOH. A precipitate forms in all three cases. Which of the following cations could the solution contain?
Both lead(II) and barium ions combine with sulfate ion and hydroxide ion to make insoluble products. But only lead (II) bromide is insoluble, while all K compounds are soluble in water.
The cation is Pb2+
Reactions and Molarity Review
1) Acetone, CH3COCH3, is a nonelectrolyte; formic acid, HCHO2, is a weak electrolyte; hypochlorous acid, HClO, is a weak electrolyte; and magnesium iodide, MgI2, is a strong electrolyte. Ammonium chloride is an electrolyte. What are the solute particles present in aqueous solutions of each compound? (Separate substances in a list with a ",".)
Acetone --> CH_3COCH_3
HCHO2 --> H^+, CHO_2^-, HCHO_2
HClO --> HClO + H^+ + ClO^-
NH4Cl- --> NH4+ + Cl-
MgI2 --> Mg^2+, I^-
Guess on the bottom part because you get 5 chances. No answer is repeated.
2) Label each of the following substances as an acid, base, salt, or none of the above. Indicate whether the substance exists in aqueous solution entirely in molecular form, entirely as ions, or as a mixture of molecules and ions.
(a) HF - acid, exists as mixture of ions and molecules
(b) acetonitrile, CH3CN - none of these, exists entirely as molecules
(c) NaClO4 - salt, exists entirely as ions
(d) Ba(OH)2 - base, exists entirely as ions
3) Which of the following are redox reactions? for those that are, indicate which element is oxidized and which is reduced. for those that are not, indicate whether they are precipitation or acid-base reactions.?
a) Cu(OH)2(s) + 2 HNO3 (aq) → Cu(NO3)2(aq) + 2 H2O (l) - It is not a redox reaction. It is acid/base neutralization reaction.
b) Fe2O3 (s) + 3 CO(g) → 2 Fe (s)+ 3 CO2(g) - It is a redox reaction. Fe3+ is reduced to Fe (zero oxidation state). C of CO is
oxidised from +2 to +4 in CO2.
c) Sr(NO3)2(aq) + H2SO4(aq) → SrSO4(s)+ 2HNO3(aq) - It is double decomposition reaction. It is not a redox reaction. SrSO4 is precipitated out.
d) 4Zn(s)+10H(aq)+2NO3(aq) → 4Zn(aq)+N2O(g) + 5H2O (l) - redox reaction - Zn is oxidized, N is reduced
e) Ca(OH)2(aq) + 2HCl(aq) → CaCl2(aq) + 2H2O(l) - acid-base reaction
f) 2 NO3‾(aq) + SO2(aq) → 2 NO2(g) + SO42-(aq) - redox reaction - S is oxidized, N is reduced
g) 2 Na(s) + 2 H2O(l) → 2 NaOH(aq) + H2(g) - redox reaction - Na is oxidized, H is reduced
4) Using the activity series (see table), write balanced chemical equations for the following reactions. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer. If no reaction occurs, enter "NONE" in the answer box.)
(a) Zinc metal is added to a solution of silver nitrate. 2AgNO_3(aq) + Zn(s) → Zn(NO_3)_2(aq) + 2Ag(s)
(b) Iron metal is added to a solution of aluminum sulfate. NONE
(c) Hydrochloric acid is added to cobalt metal. Co(s) + 2HCl(aq) → CoCl_2(aq) + H_2(g)
(d) Hydrogen gas is bubbled through an aqueous solution of FeCl2. NONE
(e) Lithium metal is added to water. 2Li(s) + 2H_2O(l) → 2LiOH(aq) + H_2(g)
5a) You have a stock solution of 14.8M NH3. how many mL of solution should you dilute to make 100.0 mL of 0.250M NH3?
M1V1=M2V2
14.8 * V1 = 0.25*100
V1 = 0.25*100/14.8
V1 = 1.689 mL
5b) If you take 10.0 mL portion of stock solution and dilute it to a total volume of 0.250 L, what would be the
concentration of the final solution?
M1V1= M2V2
14.8*10 = M2*250
M2 = 14.8*10/250
M2 = 0.592 M
6) A solution is made by mixing 13.1 g of NaOH and 76.3 mL of 0.190 M HNO3
(a) Write a balanced equation for the reaction that occurs between the solutes. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer.)
NaOH(aq) + HNO_3(aq) → H_2O(l) + NaNO_3(aq)
(b) Calculate the concentration of each ion remaining in solution. (Assume that the volume of the solution is constant.)
moles NaOH = 13.1 g/39.9968 g/mol=0.328
moles HNO3 = 0.0763 L x 0.190 M=0.0145
The greater of [H+ ] or [OH ‾ ] 0.328 - 0.0145 = 0.314 M
[Na+ ] = 0.328 / 0.0763 = 4.30 M
[NO3‾ ] = 0.190 M
(c) Is the resultant solution acidic or basic? Basic
7) A tanker truck carrying 5.3 103 kg of concentrated sulfuric acid solution tips over and spills its load. If the sulfuric acid is 95.8% H2SO4 by mass and has a density of 1.84 g/mL, how many kilograms of sodium carbonate must be added to
neutralize the acid?
5.3 x 10^3 Kg = 5.3 x 10^6 g
mass H2SO4 = 5.3 x 10^6 x 95.8 /100 = 5.08 x 10^6 g
moles H2SO4 = 5.08 x 10^6 / 98 g/mol=5.18 x 10^4
Write the balanced equation: H2SO4 + Na2CO3 = H2O + CO2 + Na2SO4
moles Na2CO3 = 5.18 x 10^4
mass Na2CO3 = 5.18 x 10^4 mol x 106 g/mol= 5.49 * 10^6 g = 5.49 x 10^3 Kg
Acetone --> CH_3COCH_3
HCHO2 --> H^+, CHO_2^-, HCHO_2
HClO --> HClO + H^+ + ClO^-
NH4Cl- --> NH4+ + Cl-
MgI2 --> Mg^2+, I^-
Guess on the bottom part because you get 5 chances. No answer is repeated.
2) Label each of the following substances as an acid, base, salt, or none of the above. Indicate whether the substance exists in aqueous solution entirely in molecular form, entirely as ions, or as a mixture of molecules and ions.
(a) HF - acid, exists as mixture of ions and molecules
(b) acetonitrile, CH3CN - none of these, exists entirely as molecules
(c) NaClO4 - salt, exists entirely as ions
(d) Ba(OH)2 - base, exists entirely as ions
3) Which of the following are redox reactions? for those that are, indicate which element is oxidized and which is reduced. for those that are not, indicate whether they are precipitation or acid-base reactions.?
a) Cu(OH)2(s) + 2 HNO3 (aq) → Cu(NO3)2(aq) + 2 H2O (l) - It is not a redox reaction. It is acid/base neutralization reaction.
b) Fe2O3 (s) + 3 CO(g) → 2 Fe (s)+ 3 CO2(g) - It is a redox reaction. Fe3+ is reduced to Fe (zero oxidation state). C of CO is
oxidised from +2 to +4 in CO2.
c) Sr(NO3)2(aq) + H2SO4(aq) → SrSO4(s)+ 2HNO3(aq) - It is double decomposition reaction. It is not a redox reaction. SrSO4 is precipitated out.
d) 4Zn(s)+10H(aq)+2NO3(aq) → 4Zn(aq)+N2O(g) + 5H2O (l) - redox reaction - Zn is oxidized, N is reduced
e) Ca(OH)2(aq) + 2HCl(aq) → CaCl2(aq) + 2H2O(l) - acid-base reaction
f) 2 NO3‾(aq) + SO2(aq) → 2 NO2(g) + SO42-(aq) - redox reaction - S is oxidized, N is reduced
g) 2 Na(s) + 2 H2O(l) → 2 NaOH(aq) + H2(g) - redox reaction - Na is oxidized, H is reduced
4) Using the activity series (see table), write balanced chemical equations for the following reactions. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer. If no reaction occurs, enter "NONE" in the answer box.)
(a) Zinc metal is added to a solution of silver nitrate. 2AgNO_3(aq) + Zn(s) → Zn(NO_3)_2(aq) + 2Ag(s)
(b) Iron metal is added to a solution of aluminum sulfate. NONE
(c) Hydrochloric acid is added to cobalt metal. Co(s) + 2HCl(aq) → CoCl_2(aq) + H_2(g)
(d) Hydrogen gas is bubbled through an aqueous solution of FeCl2. NONE
(e) Lithium metal is added to water. 2Li(s) + 2H_2O(l) → 2LiOH(aq) + H_2(g)
5a) You have a stock solution of 14.8M NH3. how many mL of solution should you dilute to make 100.0 mL of 0.250M NH3?
M1V1=M2V2
14.8 * V1 = 0.25*100
V1 = 0.25*100/14.8
V1 = 1.689 mL
5b) If you take 10.0 mL portion of stock solution and dilute it to a total volume of 0.250 L, what would be the
concentration of the final solution?
M1V1= M2V2
14.8*10 = M2*250
M2 = 14.8*10/250
M2 = 0.592 M
6) A solution is made by mixing 13.1 g of NaOH and 76.3 mL of 0.190 M HNO3
(a) Write a balanced equation for the reaction that occurs between the solutes. (Use the lowest possible coefficients. Include states-of-matter under SATP conditions in your answer.)
NaOH(aq) + HNO_3(aq) → H_2O(l) + NaNO_3(aq)
(b) Calculate the concentration of each ion remaining in solution. (Assume that the volume of the solution is constant.)
moles NaOH = 13.1 g/39.9968 g/mol=0.328
moles HNO3 = 0.0763 L x 0.190 M=0.0145
The greater of [H+ ] or [OH ‾ ] 0.328 - 0.0145 = 0.314 M
[Na+ ] = 0.328 / 0.0763 = 4.30 M
[NO3‾ ] = 0.190 M
(c) Is the resultant solution acidic or basic? Basic
7) A tanker truck carrying 5.3 103 kg of concentrated sulfuric acid solution tips over and spills its load. If the sulfuric acid is 95.8% H2SO4 by mass and has a density of 1.84 g/mL, how many kilograms of sodium carbonate must be added to
neutralize the acid?
5.3 x 10^3 Kg = 5.3 x 10^6 g
mass H2SO4 = 5.3 x 10^6 x 95.8 /100 = 5.08 x 10^6 g
moles H2SO4 = 5.08 x 10^6 / 98 g/mol=5.18 x 10^4
Write the balanced equation: H2SO4 + Na2CO3 = H2O + CO2 + Na2SO4
moles Na2CO3 = 5.18 x 10^4
mass Na2CO3 = 5.18 x 10^4 mol x 106 g/mol= 5.49 * 10^6 g = 5.49 x 10^3 Kg
Thermochemistry 1
1) Two solid objects, A and B, are placed in boiling water and allowed to come to temperature there. Each is then lifted out and placed in separate beakers containing 1000 g water at 10.0 degrees celsius. Object A increases the water temperature by 3.50 degrees celsius ; B increases the water temperature by 2.60 degrees celsius.
(a) Which object has the larger heat capacity? Object A
(b) What can you say about the specific heats of A and B? Cannot compare or determine the specific heats of the objects.
2)The specific heat of iron metal is 0.450 J/g · K. How many J of heat are necessary to raise the temperature of a 1.45-kg block of iron from 25°C to 95°C?
1.45 kg = 1450 g
mcΔt = 1450 g(.450)(95 - 25)
45675 J
3) A 2.300 g sample of quinone, C6H4O2, is burned in a bomb calorimeter whose total heat capacity is 7.854 kJ/ degrees C. The temperature of the calorimeter increases from 22.16 degrees C to 29.61 degrees C.
-What is the heat of combustion per gram of quinone?
dH = C dT
dH = (7.854 kJ/ C) ( 29.61 - 22.16)
dH = (7.854 kJ/ C) ( 7.45)
dH = 56.51kJ gained by the calorimeter, released by the combustion
56.51kJ / 2.300 grams = dH = - 25.44 kJ/gram
-What is the heat of combustion per mole of quinone?
dH = - 25.44 kJ/gram*108.1 g/mol = -2750 kJ/mol (exothermic)
(a) Which object has the larger heat capacity? Object A
(b) What can you say about the specific heats of A and B? Cannot compare or determine the specific heats of the objects.
2)The specific heat of iron metal is 0.450 J/g · K. How many J of heat are necessary to raise the temperature of a 1.45-kg block of iron from 25°C to 95°C?
1.45 kg = 1450 g
mcΔt = 1450 g(.450)(95 - 25)
45675 J
3) A 2.300 g sample of quinone, C6H4O2, is burned in a bomb calorimeter whose total heat capacity is 7.854 kJ/ degrees C. The temperature of the calorimeter increases from 22.16 degrees C to 29.61 degrees C.
-What is the heat of combustion per gram of quinone?
dH = C dT
dH = (7.854 kJ/ C) ( 29.61 - 22.16)
dH = (7.854 kJ/ C) ( 7.45)
dH = 56.51kJ gained by the calorimeter, released by the combustion
56.51kJ / 2.300 grams = dH = - 25.44 kJ/gram
-What is the heat of combustion per mole of quinone?
dH = - 25.44 kJ/gram*108.1 g/mol = -2750 kJ/mol (exothermic)
Thermochemistry 3
1) Given the data:
N2(g) + O2(g) → 2 NO(g) ΔH = +180.7 kJ
2 NO(g) + O2(g) → 2 NO2(g) ΔH = -113.1 kJ
2 N2O(g) → 2 N2(g) + O2(g) ΔH = -163.2 kJ
Use Hess's law to calculate ΔH in kJ for the reaction below:
N2O(g) + NO2(g) → 3 NO(g)
2N2O(g) → 2N2(g) + O2(g) ; divide the whole equation divided by 2 to get N2O(g) → N2(g) + 1/2O2(g)
2NO2(g) → 2NO(g) + O2(g) ; divide the reverse of the second equation by 2 to get NO2(g) → NO(g) + 1/2O2(g)
N2(g) + O2(g) → 2NO(g)
Add them up, and you get (Equation 3 divided by 2) + (Reverse of Equation 2 divided by 2) + Equation 1:
N2O(g) → N2(g) + 1/2O2(g)
NO2(g) → NO(g) + 1/2O2(g)
N2(g) + O2(g) → 2NO(g)
=======================
N2O(g) + NO2(g) → 3NO(g)
Numerically, ΔH = (-163.2/2) + (113.1/2) + (180.7) = +155.65 kJ
2) Calculate the value of ΔH° for each of the following reactions.
According to Hess' law enthalpy of reaction equals difference of enthalpy of formation of products and reactants
(a) 3 Fe(s) + 4 CO2(g) → 4 CO(g) + Fe3O4(s)
ΔH° = kJ
ΔHr° = 4*ΔHf°(CO(g)) + ΔHf°(Fe3O4(s)) - 3*ΔHf°(Fe(s)) - 4*ΔHf°(CO₂(g))
= 4*(-110.53kJ) + (-1120.89kJ) - 3·0 - 4*ΔHf°(-393.52kJ)
= 11.07kJ
(b) 4 NH3(g) + O2(g) → 2 N2H4(g) + 2 H2O(l)
ΔH° = kJ
ΔHr° = 2·ΔHf°(N₂H₄(g)) + 2·ΔHf°(H2O(l)) - 4·ΔHf°(NH₃(g)) - ΔHf°(O₂(g))
= 2*(95.35kJ) + 2*(-285.83kJ) - 4*(-45.94kJ) - 0
= -197.2kJ
(c) 6 C(s) + 6 H2O(l) → C6H12O6(s)
ΔH° = kJ
ΔHr° = ΔHf°(C6H12O6(s)) - 6·ΔHf°(C(s)) - 6·ΔHf°(H2O(l))
= -1274.5kJ - 6*0 - 6*(-285.83kJ)
= +440.48kJ
(d) Fe2O3(s) + 6 HCl(g) → 2 FeCl3(s) + 3 H2O(g)
ΔH° = kJ
ΔHr° = 2·ΔHf°(FeCl₃(s)) + 3·ΔHf°(H₂O(g)) - ΔHf°(Fe₂O₃(s)) - 6·ΔHf°(HCl(g))
= 2*(-399.41) + 3*ΔHf°(-241.83kJ) - (-825.50kJ) - 6*(-92.31kJ)
= -144.95kJ
3) Consider the following reaction. 2Mg(s) + O2(g) → 2MgO(s) ΔH = −1204 kJ
(a) Is this reaction exothermic or endothermic? exothermic, because enthalpy (ΔH) is negative
(b) Calculate the amount of heat transferred when 2.5 g of Mg(s) reacts at constant pressure.
Moles * formula weight = grams.
The balanced equation indicates that 2 moles of Mg (48.6 g) react with one mole of O2 (32.0 g) to produce 2 moles of MgO (80.6 g). Using those amounts, the reaction gives off 1204 kJ of heat. The first question asks what happens when we change the amount of Mg to something other than 2 moles, like 2.5 g.
2.5 g Mg x (1 mole Mg / 24.3 g)
x (-1204 kJ / 2 moles Mg) = -61.934 kJ
(c) How many grams of MgO are produced during an enthalpy change of 96.0 kJ?
96 kJ x (2 moles MgO / -1204 kJ) x (40.3 g MgO / 1 mole MgO) = 6.43 g MgO
(d) How many kilojoules of heat are absorbed when 7.5 g of MgO(s) are decomposed into Mg(s) and O2(g) at constant pressure?
If 2 moles of MgO (which is 80.6 g) require 1204 kJ to decompose, how much heat will 7.5 g of MgO need?
7.5 g MgO x (1 mole MgO / 40.3 g MgO) x (1204 kJ / 2 moles MgO) = 112 kJ
4)Many cigarette lighters contain liquid butane, C4H10(l). Using enthalpies of formation, calculate the quantity of heat produced when 3.30 g of butane is completely combusted in air under standard conditions.
Enthalpies of formation apply to the individual chemical bonds. Firstly you need to determine the chemical reaction:
1 mol of C4H10 needs to produce 4 mols of CO2 and 5 mols of H2O. If you now count up how many mols
of O are required then this comes to 8+5=13. Oxygen molecules have 2 O atoms so to balance this equation you get:
2C4H10+13O2 --> 8CO2 + 10H2O
Now you need to apply the Enthalpies of each of the four different molecules. Everything on the left is heat consumption or negative heat release and everything on the right is heat release. So now get your tables out that define the enthalpies. C4H10 = 30.1 Kcal/mol
CO2 = 393.5 Kcal/mol
H2O = 285.5 Kcal/mol
O2 is neutral.
Now work out how many mols you have. Molecular weight of butane is 58 so 3.3/58 gives 0.0568 mols.
Now you can do the calculation to work out heat release. On the left (pre-combustion) you have 0.0568x30.1=1.71 Kcals
On the right you have 4x0.0568x393.5 + 5x0.0568x285.5 = 170.78Kcal
Deduct the left from the right and that is the total heat release =170.78-1.71 = 169 kcal
1a) What is the frequency of radiation that has a wavelength of 0.952 µm?
λ = hc/v
c = speed of light (3*10^8)
h = planks
constant
v = frequency
λ is your wavelength
Plank's constant = 6.626*10^-34
So ((6.626*10^-34) * (3*10^8)) / (0,952*10^-6m) = wavelength
1b) What is the wavelength of radiation that has a frequency of 5.60×10^14 s^-1?
C = speed of
light = 3 x 10^8 m/s
wavelength
is usually expressed in nm
C = wavelength x frequency
C = lambda x nu
(3 x 10^8 m/s) / (5.6 x 10^14 /s) = 5.35 x 10^-7 m
there are 1 x 10^9 nm / 1 m
(5.35 x 10^-7) x (1 x 10^9nm/1m) = 535.7 nm
1c) Would the radiations in part (a) or part (b) be visible to the human eye?
The radiation in part (b) would be visible.
1d) What distance does electromagnetic radiation travel in 19 µs?
EM radiation is light. The speed of light is about 3.0 X 10^8 meters per second. 19 us = 19 * 10^-6 seconds
(3.0* 10^8 m/s) * (19 *10^-6 s) = 5.7* 10^3 m = 5700 m
2) An AM radio station broadcasts at 1540 kHz, and its FM partner broadcasts at 101.0 MHz. Calculate and compare the energy of photons emitted by these radio stations.
E=hf
h= planck's constant = 6.626068 × 10^-34 m2 kg / s
so E1= h* 1540000 Hz
AM= 1.0204 *(10^-27)
E2= h* 101000000 hertz
FM= 6.69*(10^-26)
EFM / EAM = [1.0204 *(10^-27)] / [6.69*(10^-26)]
3) Among the elementary subatomic particles of physics is the muon, which decays within a few nanoseconds after formation. The muon has a rest mass 206.8 times that of an electron. Calculate the de Broglie wavelength associated with a
muon traveling at a velocity of 8.70×10^5 cm/s.
velocity = 8.70x10^3 m/sec
mass = 206.8x9.1083x10^-31 kg = 1.884x10^-28 kg
Plank's constant (h) = 6.63x10^-34 kg m^2/sec
wavelength = 6.63x10^-34 / (8.70x10^3 x 1.884x10^-28 kg)
wavelength = 4.04x10^-10 m
Convert this wavelength (in meters) to the desired units.
units: kg m^2/sec / (kg x m/sec) = m
Plank's constant = 6.63x10^-34 J sec
1 J = 1 kg m^2/sec^2
Plank's constant = 6.63x10^-34 kg m^2/sec
4) For each of the following electronic transitions in the hydrogen atom, calculate the energy, frequency, and wavelength of the associated radiation, and determine whether the radiation is emitted or absorbed during the transition.
(a) from n = 5 to n = 2
energy = -4.578e-19 J
frequency = (4.578e-19)/(6.626e-34) s−1
wavelength = -(6.626e-34)*(3e8)/(-4.578e-19) m
The radiation is emitted during the transition.
N2(g) + O2(g) → 2 NO(g) ΔH = +180.7 kJ
2 NO(g) + O2(g) → 2 NO2(g) ΔH = -113.1 kJ
2 N2O(g) → 2 N2(g) + O2(g) ΔH = -163.2 kJ
Use Hess's law to calculate ΔH in kJ for the reaction below:
N2O(g) + NO2(g) → 3 NO(g)
2N2O(g) → 2N2(g) + O2(g) ; divide the whole equation divided by 2 to get N2O(g) → N2(g) + 1/2O2(g)
2NO2(g) → 2NO(g) + O2(g) ; divide the reverse of the second equation by 2 to get NO2(g) → NO(g) + 1/2O2(g)
N2(g) + O2(g) → 2NO(g)
Add them up, and you get (Equation 3 divided by 2) + (Reverse of Equation 2 divided by 2) + Equation 1:
N2O(g) → N2(g) + 1/2O2(g)
NO2(g) → NO(g) + 1/2O2(g)
N2(g) + O2(g) → 2NO(g)
=======================
N2O(g) + NO2(g) → 3NO(g)
Numerically, ΔH = (-163.2/2) + (113.1/2) + (180.7) = +155.65 kJ
2) Calculate the value of ΔH° for each of the following reactions.
According to Hess' law enthalpy of reaction equals difference of enthalpy of formation of products and reactants
(a) 3 Fe(s) + 4 CO2(g) → 4 CO(g) + Fe3O4(s)
ΔH° = kJ
ΔHr° = 4*ΔHf°(CO(g)) + ΔHf°(Fe3O4(s)) - 3*ΔHf°(Fe(s)) - 4*ΔHf°(CO₂(g))
= 4*(-110.53kJ) + (-1120.89kJ) - 3·0 - 4*ΔHf°(-393.52kJ)
= 11.07kJ
(b) 4 NH3(g) + O2(g) → 2 N2H4(g) + 2 H2O(l)
ΔH° = kJ
ΔHr° = 2·ΔHf°(N₂H₄(g)) + 2·ΔHf°(H2O(l)) - 4·ΔHf°(NH₃(g)) - ΔHf°(O₂(g))
= 2*(95.35kJ) + 2*(-285.83kJ) - 4*(-45.94kJ) - 0
= -197.2kJ
(c) 6 C(s) + 6 H2O(l) → C6H12O6(s)
ΔH° = kJ
ΔHr° = ΔHf°(C6H12O6(s)) - 6·ΔHf°(C(s)) - 6·ΔHf°(H2O(l))
= -1274.5kJ - 6*0 - 6*(-285.83kJ)
= +440.48kJ
(d) Fe2O3(s) + 6 HCl(g) → 2 FeCl3(s) + 3 H2O(g)
ΔH° = kJ
ΔHr° = 2·ΔHf°(FeCl₃(s)) + 3·ΔHf°(H₂O(g)) - ΔHf°(Fe₂O₃(s)) - 6·ΔHf°(HCl(g))
= 2*(-399.41) + 3*ΔHf°(-241.83kJ) - (-825.50kJ) - 6*(-92.31kJ)
= -144.95kJ
3) Consider the following reaction. 2Mg(s) + O2(g) → 2MgO(s) ΔH = −1204 kJ
(a) Is this reaction exothermic or endothermic? exothermic, because enthalpy (ΔH) is negative
(b) Calculate the amount of heat transferred when 2.5 g of Mg(s) reacts at constant pressure.
Moles * formula weight = grams.
The balanced equation indicates that 2 moles of Mg (48.6 g) react with one mole of O2 (32.0 g) to produce 2 moles of MgO (80.6 g). Using those amounts, the reaction gives off 1204 kJ of heat. The first question asks what happens when we change the amount of Mg to something other than 2 moles, like 2.5 g.
2.5 g Mg x (1 mole Mg / 24.3 g)
x (-1204 kJ / 2 moles Mg) = -61.934 kJ
(c) How many grams of MgO are produced during an enthalpy change of 96.0 kJ?
96 kJ x (2 moles MgO / -1204 kJ) x (40.3 g MgO / 1 mole MgO) = 6.43 g MgO
(d) How many kilojoules of heat are absorbed when 7.5 g of MgO(s) are decomposed into Mg(s) and O2(g) at constant pressure?
If 2 moles of MgO (which is 80.6 g) require 1204 kJ to decompose, how much heat will 7.5 g of MgO need?
7.5 g MgO x (1 mole MgO / 40.3 g MgO) x (1204 kJ / 2 moles MgO) = 112 kJ
4)Many cigarette lighters contain liquid butane, C4H10(l). Using enthalpies of formation, calculate the quantity of heat produced when 3.30 g of butane is completely combusted in air under standard conditions.
Enthalpies of formation apply to the individual chemical bonds. Firstly you need to determine the chemical reaction:
1 mol of C4H10 needs to produce 4 mols of CO2 and 5 mols of H2O. If you now count up how many mols
of O are required then this comes to 8+5=13. Oxygen molecules have 2 O atoms so to balance this equation you get:
2C4H10+13O2 --> 8CO2 + 10H2O
Now you need to apply the Enthalpies of each of the four different molecules. Everything on the left is heat consumption or negative heat release and everything on the right is heat release. So now get your tables out that define the enthalpies. C4H10 = 30.1 Kcal/mol
CO2 = 393.5 Kcal/mol
H2O = 285.5 Kcal/mol
O2 is neutral.
Now work out how many mols you have. Molecular weight of butane is 58 so 3.3/58 gives 0.0568 mols.
Now you can do the calculation to work out heat release. On the left (pre-combustion) you have 0.0568x30.1=1.71 Kcals
On the right you have 4x0.0568x393.5 + 5x0.0568x285.5 = 170.78Kcal
Deduct the left from the right and that is the total heat release =170.78-1.71 = 169 kcal
1a) What is the frequency of radiation that has a wavelength of 0.952 µm?
λ = hc/v
c = speed of light (3*10^8)
h = planks
constant
v = frequency
λ is your wavelength
Plank's constant = 6.626*10^-34
So ((6.626*10^-34) * (3*10^8)) / (0,952*10^-6m) = wavelength
1b) What is the wavelength of radiation that has a frequency of 5.60×10^14 s^-1?
C = speed of
light = 3 x 10^8 m/s
wavelength
is usually expressed in nm
C = wavelength x frequency
C = lambda x nu
(3 x 10^8 m/s) / (5.6 x 10^14 /s) = 5.35 x 10^-7 m
there are 1 x 10^9 nm / 1 m
(5.35 x 10^-7) x (1 x 10^9nm/1m) = 535.7 nm
1c) Would the radiations in part (a) or part (b) be visible to the human eye?
The radiation in part (b) would be visible.
1d) What distance does electromagnetic radiation travel in 19 µs?
EM radiation is light. The speed of light is about 3.0 X 10^8 meters per second. 19 us = 19 * 10^-6 seconds
(3.0* 10^8 m/s) * (19 *10^-6 s) = 5.7* 10^3 m = 5700 m
2) An AM radio station broadcasts at 1540 kHz, and its FM partner broadcasts at 101.0 MHz. Calculate and compare the energy of photons emitted by these radio stations.
E=hf
h= planck's constant = 6.626068 × 10^-34 m2 kg / s
so E1= h* 1540000 Hz
AM= 1.0204 *(10^-27)
E2= h* 101000000 hertz
FM= 6.69*(10^-26)
EFM / EAM = [1.0204 *(10^-27)] / [6.69*(10^-26)]
3) Among the elementary subatomic particles of physics is the muon, which decays within a few nanoseconds after formation. The muon has a rest mass 206.8 times that of an electron. Calculate the de Broglie wavelength associated with a
muon traveling at a velocity of 8.70×10^5 cm/s.
velocity = 8.70x10^3 m/sec
mass = 206.8x9.1083x10^-31 kg = 1.884x10^-28 kg
Plank's constant (h) = 6.63x10^-34 kg m^2/sec
wavelength = 6.63x10^-34 / (8.70x10^3 x 1.884x10^-28 kg)
wavelength = 4.04x10^-10 m
Convert this wavelength (in meters) to the desired units.
units: kg m^2/sec / (kg x m/sec) = m
Plank's constant = 6.63x10^-34 J sec
1 J = 1 kg m^2/sec^2
Plank's constant = 6.63x10^-34 kg m^2/sec
4) For each of the following electronic transitions in the hydrogen atom, calculate the energy, frequency, and wavelength of the associated radiation, and determine whether the radiation is emitted or absorbed during the transition.
(a) from n = 5 to n = 2
energy = -4.578e-19 J
frequency = (4.578e-19)/(6.626e-34) s−1
wavelength = -(6.626e-34)*(3e8)/(-4.578e-19) m
The radiation is emitted during the transition.
Light II
1 (a)
What is the frequency of radiation whose wavelength is 20.75 Å?
(2.9979×10^8 m/s) / (20.75×10^(-10) m) = 1.445×10^17 sˉ¹.
(b) What is the wavelength of radiation that has a frequency of 2.6×10^12 s-1?
(2.9979×10^8 m/s) / (2.6×10^12 sˉ¹) = 1.15×10^(-4) m.
(c) Would the radiations in part (a) or part (b) be detected by an X-ray detector? (Select all that apply.)
The radiation in part (a) would be detected.
(d) What distance does electromagnetic radiation travel in 13 fs?
(2.9979×10^8 m/s)(13×10^(-15) s) = 3.9×10^(-6) m.
2 (a) Calculate the energy of photons with wavelength 3.2 µm and 0.161 nm.
E = hc/λ
3.2 µm photon: E = (6.63×10^(-34) J·s)(3×10^8 m/s) / (3.2×10^(-6) m) = Answer
0.161 nm photon: E = (6.63×10^(-34) J·s)(3×10^8 m/s) / (0.161×10^(-9) m) = Answer
(b) Identify the region of the electromagnetic spectrum to which each belongs.
3.2 µm photon: microwaves
0.161 nm photon: x-rays
3.6 µm photon: infared
.157 nm photom: x-rays
3) Use the de Broglie relationship, λ = h/mv, to determine the wavelengths of the following objects.
(a) An 85-kg person skiing at 50 km/hr
λ = h/mv
6.63 * 10e-34 J-s / (85 kg * 50 km/hr)
x (1 kg-m/s^2) / (1 J)
x (3600 s) / (1 hr)
x (1 km) / (1000 m)
= 5e-40m
(b) 10.5 g golf ball traveling at 390. m/s
the velocity is already in m/s, so just convert mass into kg by dividing by 1000.
(c) lithium atom moving at 2.6×10^5 m/s
The atomic weight of Lithium is 6.9 g/mole. If we divide by 1000, to get to kg and then by Avogadro's number, we get
m=1.15*10^-25
λ =h/mv = (6.63*10^-34)/[(1.15*10^-25)(2.6×10^5)] = 2.2*10^-14 meters
4) Indicate whether energy is emitted or absorbed when the following electronic transitions occur in hydrogen.
When levels go from a lower "n" to a higher "n," energy is absorbed. When levels go from a higher"n" to a higher "n," energy is emitted. A radius of (example) 5Å to 1Å still applies. This example would have energy emitted.
5) The Lyman series of emission lines of the hydrogen atom are those for which nf = 1.
(a) Determine the region of the electromagnetic spectrum in which the lines of the Lyman series are observed. ultraviolet
(b) Calculate the wavelengths of the first three lines in the Lyman series—those for which ni = 2, 3, and 4. (Enter your answers to three digits.)
ni = 2
1.21e-7 m
ni = 3
1.03e-7 m
ni = 4
9.72e-8 m
What is the frequency of radiation whose wavelength is 20.75 Å?
(2.9979×10^8 m/s) / (20.75×10^(-10) m) = 1.445×10^17 sˉ¹.
(b) What is the wavelength of radiation that has a frequency of 2.6×10^12 s-1?
(2.9979×10^8 m/s) / (2.6×10^12 sˉ¹) = 1.15×10^(-4) m.
(c) Would the radiations in part (a) or part (b) be detected by an X-ray detector? (Select all that apply.)
The radiation in part (a) would be detected.
(d) What distance does electromagnetic radiation travel in 13 fs?
(2.9979×10^8 m/s)(13×10^(-15) s) = 3.9×10^(-6) m.
2 (a) Calculate the energy of photons with wavelength 3.2 µm and 0.161 nm.
E = hc/λ
3.2 µm photon: E = (6.63×10^(-34) J·s)(3×10^8 m/s) / (3.2×10^(-6) m) = Answer
0.161 nm photon: E = (6.63×10^(-34) J·s)(3×10^8 m/s) / (0.161×10^(-9) m) = Answer
(b) Identify the region of the electromagnetic spectrum to which each belongs.
3.2 µm photon: microwaves
0.161 nm photon: x-rays
3.6 µm photon: infared
.157 nm photom: x-rays
3) Use the de Broglie relationship, λ = h/mv, to determine the wavelengths of the following objects.
(a) An 85-kg person skiing at 50 km/hr
λ = h/mv
6.63 * 10e-34 J-s / (85 kg * 50 km/hr)
x (1 kg-m/s^2) / (1 J)
x (3600 s) / (1 hr)
x (1 km) / (1000 m)
= 5e-40m
(b) 10.5 g golf ball traveling at 390. m/s
the velocity is already in m/s, so just convert mass into kg by dividing by 1000.
(c) lithium atom moving at 2.6×10^5 m/s
The atomic weight of Lithium is 6.9 g/mole. If we divide by 1000, to get to kg and then by Avogadro's number, we get
m=1.15*10^-25
λ =h/mv = (6.63*10^-34)/[(1.15*10^-25)(2.6×10^5)] = 2.2*10^-14 meters
4) Indicate whether energy is emitted or absorbed when the following electronic transitions occur in hydrogen.
When levels go from a lower "n" to a higher "n," energy is absorbed. When levels go from a higher"n" to a higher "n," energy is emitted. A radius of (example) 5Å to 1Å still applies. This example would have energy emitted.
5) The Lyman series of emission lines of the hydrogen atom are those for which nf = 1.
(a) Determine the region of the electromagnetic spectrum in which the lines of the Lyman series are observed. ultraviolet
(b) Calculate the wavelengths of the first three lines in the Lyman series—those for which ni = 2, 3, and 4. (Enter your answers to three digits.)
ni = 2
1.21e-7 m
ni = 3
1.03e-7 m
ni = 4
9.72e-8 m
Electrons & Quantum Numbers I
1(a) For n = 4, what are the possible values of L and mL?
L = 0,1,2,3 (n-1=4-1=3)
When L = 0 ml =0
when L = 1 ml = -1 , 0 , +1
when L = 2 ml = -2 , -1 , 0 , +1 , +2
when L = 3 ml = -3 , - 2 , -1 , 0, +1 , +2 , +3
(b) For L = 2, what are the possible values of mL?
-2,-1,0,1,2
2) Select all of the following quantum number sets that are allowed for the most energetic
electron in chlorine. (Hint: Write the electron configuration for chlorine first.)
(3,1,1,1/2)
(3,1,-1,-1/2)
(3,1,-1,1/2)
(3,1,1,-1/2)
(3,1,0,-1/2)
(3,1,0,1/2)
3) Sellect all of the following quantum number sets that are not allowed?
(2,1,-2,1/2)
(2,0,-1,-3/2)
(1,1,1,1/2)
(3,2,-3,1/2)
Mark all of the following quantum numbers that are allowed?
(2,1,-1,1/2)
(2,0,0,1/2)
(1,0,0,1/2)
4) Write the condensed electron configurations for the following atoms, and indicate how many unpaired electrons each has. http://en.wikipedia.org/wiki/Electron_configurations_of_the_elements_(data_page)
5) Identify the specific element that corresponds to each of the following electron configurations.
http://en.wikipedia.org/wiki/Electron_configurations_of_the_elements_(data_page)
6) (a) How many possible values for l and ml are there when n = 2?
number of values for l
2
total number of values for ml
2^2 = 4
(b)How many possible values for l and ml are there when n = 8?
number of values for l
8
total number of values for ml
8^2 = 64
7) Which of the following represent impossible combinations of n and ?
Look at Periodic Table of Elements
8) What is the maximum number of electrons in an atom that can have the following quantum numbers?
n is the principal quantum energy level and has values 1,2,3...
l has integral values from 0 to n-1.
m_l has integral values from -l to +l.
m_s has values +1/2 or -1/2.
1) n=2, m_s=1/2
for l=0 there is only m_l =0 and m_s= +1/2, -1/2
for l=1 there are only m_l=-1,0,1 and m_s=+1/2,-1/2
thus, there are a total of 4 electrons that have these quantum numbers.
2) n=5, l=3
If l=3 then m_l = -3,-2,-1,0,1,2,3.
For each orbital, there are two electrons, m_s=+1/2,-1/2
thus, there are a total of 14 electrons that have these quantum numbers.
3) n=4, l=3, m_l=-3
This specifies a particular orbital which can contain electrons. Since each orbital
can contain a maximum of 2 electron, there are a total of 2 electrons that can
have these quantum numbers.
L = 0,1,2,3 (n-1=4-1=3)
When L = 0 ml =0
when L = 1 ml = -1 , 0 , +1
when L = 2 ml = -2 , -1 , 0 , +1 , +2
when L = 3 ml = -3 , - 2 , -1 , 0, +1 , +2 , +3
(b) For L = 2, what are the possible values of mL?
-2,-1,0,1,2
2) Select all of the following quantum number sets that are allowed for the most energetic
electron in chlorine. (Hint: Write the electron configuration for chlorine first.)
(3,1,1,1/2)
(3,1,-1,-1/2)
(3,1,-1,1/2)
(3,1,1,-1/2)
(3,1,0,-1/2)
(3,1,0,1/2)
3) Sellect all of the following quantum number sets that are not allowed?
(2,1,-2,1/2)
(2,0,-1,-3/2)
(1,1,1,1/2)
(3,2,-3,1/2)
Mark all of the following quantum numbers that are allowed?
(2,1,-1,1/2)
(2,0,0,1/2)
(1,0,0,1/2)
4) Write the condensed electron configurations for the following atoms, and indicate how many unpaired electrons each has. http://en.wikipedia.org/wiki/Electron_configurations_of_the_elements_(data_page)
5) Identify the specific element that corresponds to each of the following electron configurations.
http://en.wikipedia.org/wiki/Electron_configurations_of_the_elements_(data_page)
6) (a) How many possible values for l and ml are there when n = 2?
number of values for l
2
total number of values for ml
2^2 = 4
(b)How many possible values for l and ml are there when n = 8?
number of values for l
8
total number of values for ml
8^2 = 64
7) Which of the following represent impossible combinations of n and ?
Look at Periodic Table of Elements
8) What is the maximum number of electrons in an atom that can have the following quantum numbers?
n is the principal quantum energy level and has values 1,2,3...
l has integral values from 0 to n-1.
m_l has integral values from -l to +l.
m_s has values +1/2 or -1/2.
1) n=2, m_s=1/2
for l=0 there is only m_l =0 and m_s= +1/2, -1/2
for l=1 there are only m_l=-1,0,1 and m_s=+1/2,-1/2
thus, there are a total of 4 electrons that have these quantum numbers.
2) n=5, l=3
If l=3 then m_l = -3,-2,-1,0,1,2,3.
For each orbital, there are two electrons, m_s=+1/2,-1/2
thus, there are a total of 14 electrons that have these quantum numbers.
3) n=4, l=3, m_l=-3
This specifies a particular orbital which can contain electrons. Since each orbital
can contain a maximum of 2 electron, there are a total of 2 electrons that can
have these quantum numbers.
More light practice & photoelectric effect
1) A diode laser emits at a wavelength of 987 nm.
(a) In what portion of the electromagnetic spectrum is this radiation found?
Infared
(b) All of its output energy is absorbed in a detector that measures a total energy of 0.47 J over a period of 38 sec. How many photons per second are being emitted by the laser?
Absorption per second is 0.47/38 = 0.0124 J/s.
Energy per photon is hc/λ = (6.63×10^(-34) J·s)(2.9979×10^8 m/s) / (987×10^-9 m) = 2.0×10^-19 J.
So, (0.0124 J/s) / (2.0×10^(-19) J/photon) = 6.1×10^16 photons/s.
2) A stellar object is emitting radiation at 1220 nm.
(a) What type of electromagnetic spectrum is this radiation?
infrared
(b) If the detector is capturing 9x10^7 photons per second at this wavelength, what is the total energy of the photons detected in one hour?
E = hc/λ = (6.63×10^(-34) J·s)(2.9979×10^8 m/s) / (1220×10^(-9) m) = 1.63×10^(-19) J/photon.
Total energy = (1.63×10^(-19) J/photon)(9×10^7 photons/s)(3600 s/hr) = 5.28×10^(-8) J/hr
3) Molybdenum metal must absorb radiation with a minimum frequency of 1.09 10^15 s−1 before it can eject an electron from its surface via the photoelectric effect.
(a) What is the minimum energy needed to eject an electron?
Threshold frequency (v0)=1.09 ×10^15 s^-1
The minimum energy required to emit a photoelectron from the surface of a metal is equal to hv0, where h is the
Planck's constant (6.626068 × 10^-34 J s), and v0 is the threshold frequency. This energy is known as the threshold energy of the metal.
Minimum energy required to emit a photoelectron from the surface of Molybdenum
energy=hv0=6.62×10^-34 ×(1.09 ×10^15) = 7.22×10^-19 J
(b) What wavelength of radiation will provide a photon of this energy?
λ=c/v0= (3×10^8)/(1.09 ×10^15) m = 2.752×10^-7 m
2.752×10^-7 m * 10^9 = 275 nm
(c) If molybdenum is irradiated with light of wavelength of 174 nm, what is the maximum possible kinetic energy of the emitted electrons?
λ=120 nm=120 ×10^-9 m=1.2 × 10^-7. Therefore, frequency (v)=c/λ= ( 3×10^8)/(1.2 × 10^-7)= 2.5 × 10^15 Hz
Emax=hv-hv0=h(v-v0)=6.63×10^-34 [(2.5 × 10^15 ) - (1.09 ×10^15)] = 9.34×10^-19 J
4) Sodium requires a photon with a minimum energy of 4.41 10-19 J to emit electrons.
(a) What is the minimum frequency of light necessary to emit electrons from sodium via the photoelectric effect?
Planck's constant: h = 6.63 x 10^(-34) J/s
Frequency ν = E/h = 4.41 x 10^(-19)/h = 6.656 x 10^(14)
(b) What is the wavelength of this light?
Speed of light c = 3 x 10^8 m/s
λ = c/6.656 x 10(14) = 4.504 x 10^(-7) m = 450.4 nm
(c) If sodium is irradiated with light of 402 nm, what is the maximum possible kinetic energy of the emitted electrons?
Energy of photons of wavelength 402 nm = hc/402 x 10^(-9) = 4.941 x 10^(-19) J.
Maximum possible kinetic energy of emitted electrons = (4.941 - 4.41) x 10^(-19) = 5.31 x 10^(-20) J.
(d) What is the maximum number of electrons that can be freed by a burst of light (λ = 402 nm) whose total energy is 1.28 µJ?
number of electrons = [ 1.28E-6 ] / [ 10^9 * h * c / L ]
number of electrons = [ 1.28E-6 ] / [ 10^9 * 6.626 068 96E-34 * 299 792 458 / 402 ]
number of electrons = 2.59E12
(a) In what portion of the electromagnetic spectrum is this radiation found?
Infared
(b) All of its output energy is absorbed in a detector that measures a total energy of 0.47 J over a period of 38 sec. How many photons per second are being emitted by the laser?
Absorption per second is 0.47/38 = 0.0124 J/s.
Energy per photon is hc/λ = (6.63×10^(-34) J·s)(2.9979×10^8 m/s) / (987×10^-9 m) = 2.0×10^-19 J.
So, (0.0124 J/s) / (2.0×10^(-19) J/photon) = 6.1×10^16 photons/s.
2) A stellar object is emitting radiation at 1220 nm.
(a) What type of electromagnetic spectrum is this radiation?
infrared
(b) If the detector is capturing 9x10^7 photons per second at this wavelength, what is the total energy of the photons detected in one hour?
E = hc/λ = (6.63×10^(-34) J·s)(2.9979×10^8 m/s) / (1220×10^(-9) m) = 1.63×10^(-19) J/photon.
Total energy = (1.63×10^(-19) J/photon)(9×10^7 photons/s)(3600 s/hr) = 5.28×10^(-8) J/hr
3) Molybdenum metal must absorb radiation with a minimum frequency of 1.09 10^15 s−1 before it can eject an electron from its surface via the photoelectric effect.
(a) What is the minimum energy needed to eject an electron?
Threshold frequency (v0)=1.09 ×10^15 s^-1
The minimum energy required to emit a photoelectron from the surface of a metal is equal to hv0, where h is the
Planck's constant (6.626068 × 10^-34 J s), and v0 is the threshold frequency. This energy is known as the threshold energy of the metal.
Minimum energy required to emit a photoelectron from the surface of Molybdenum
energy=hv0=6.62×10^-34 ×(1.09 ×10^15) = 7.22×10^-19 J
(b) What wavelength of radiation will provide a photon of this energy?
λ=c/v0= (3×10^8)/(1.09 ×10^15) m = 2.752×10^-7 m
2.752×10^-7 m * 10^9 = 275 nm
(c) If molybdenum is irradiated with light of wavelength of 174 nm, what is the maximum possible kinetic energy of the emitted electrons?
λ=120 nm=120 ×10^-9 m=1.2 × 10^-7. Therefore, frequency (v)=c/λ= ( 3×10^8)/(1.2 × 10^-7)= 2.5 × 10^15 Hz
Emax=hv-hv0=h(v-v0)=6.63×10^-34 [(2.5 × 10^15 ) - (1.09 ×10^15)] = 9.34×10^-19 J
4) Sodium requires a photon with a minimum energy of 4.41 10-19 J to emit electrons.
(a) What is the minimum frequency of light necessary to emit electrons from sodium via the photoelectric effect?
Planck's constant: h = 6.63 x 10^(-34) J/s
Frequency ν = E/h = 4.41 x 10^(-19)/h = 6.656 x 10^(14)
(b) What is the wavelength of this light?
Speed of light c = 3 x 10^8 m/s
λ = c/6.656 x 10(14) = 4.504 x 10^(-7) m = 450.4 nm
(c) If sodium is irradiated with light of 402 nm, what is the maximum possible kinetic energy of the emitted electrons?
Energy of photons of wavelength 402 nm = hc/402 x 10^(-9) = 4.941 x 10^(-19) J.
Maximum possible kinetic energy of emitted electrons = (4.941 - 4.41) x 10^(-19) = 5.31 x 10^(-20) J.
(d) What is the maximum number of electrons that can be freed by a burst of light (λ = 402 nm) whose total energy is 1.28 µJ?
number of electrons = [ 1.28E-6 ] / [ 10^9 * h * c / L ]
number of electrons = [ 1.28E-6 ] / [ 10^9 * 6.626 068 96E-34 * 299 792 458 / 402 ]
number of electrons = 2.59E12
Periodic Trends II
1) Arrange the following atoms in order of increasing effective nuclear charge experienced by the electrons in the n = 3 electron shell. (Use the appropriate <, =, or > symbol to separate substances in the list.)
Effective nuclear charge increases going down and across (from left to right). Effective nuclear charge is the charge felt by valence electrons of an atom after you have taken into account the number of shielding electrons that surround the nucleus.
IF YOU WANT TO LEARN THIS READ BELOW.
The effective nuclear charge (Zeff) is calculated by knowing the atomic number of the atom (Z, number of protons), and the number of electrons that lie in shells closer to the nucleus than the electron you're looking at. This second part is
the shielding (S) that the electron of interest experiences. The overall calculation is: Zeff = Z - S. Determining Z is easy--just look up the atomic number of the element (it's on the periodic table). Figuring out S is also easy. The value of S is equal to the number of electrons that lie in shells with n values smaller than the n for the electron you're interested in. So, if you are calculating the shielding for a 3p electron, then all electrons with n values of 1 or 2 contribute to the shielding; this would be 2 (n=1) + 8 (n=2) = 10 total electrons, or S = 10.
Explain the basis for your order.
The shielding of electrons in the n=3 shell by 1s and 2s core electrons in these elements is approximately equal, so the effective nuclear charge increases as Z increases.
2a) How do the sizes of atoms change as we move from left to right across a row in the periodic table?
Size decreases
b) How do the sizes of atoms change as we move from top to bottom in a group in the periodic table?
Size increases
c) Arrange the following atoms in order of increasing atomic radius: C, I, Rb, and F. (Use the appropriate <, =, or > symbol to separate substances in the list.) Refer to 2a) and 2b)
F < C < I < Rb
3a) Among the nonmetallic elements, the change in atomic radius in moving one place left or right in a row is smaller than the change in moving one row up or down. Explain these observations.
A unit change in principal quantum number has a much larger effect on the size of the outermost orbital than the change in electrostatic attraction between the outer electrons and a nucleus with one more or one less proton
4 a,b,c) Refer to 2a) and 2b)
5) Write equations that show the processes that describe the first, second, and third ionization energies of a boron atom. (Omit states-of-matter in your answer.)
I1 = B --> B^+ + e^-
I2 = B^+ --> B^2+ + e^-
I3 = B^2+ --> B^3+ + e^-
6) For each of the following pairs, indicate which element has the larger first ionization energy.
Ionization energy increases going up and across (from left to right).
7) For each of the following pairs, which element will have the greater metallic character?
Use periodic table: more metallic to the left and lower down basically, as this makes it easier to form positive ions - the characterisitic of a good metal
1) Sb or As As is less metallic (higher up then Sb, both metalloids though)
2) Be or Na Be (higher and further to the right than Na, but both metals)
3) Si or P P (futher to right P is non metal Si is metalloid)
4) Cs or Au Au (further to right, but both metals)
Effective nuclear charge increases going down and across (from left to right). Effective nuclear charge is the charge felt by valence electrons of an atom after you have taken into account the number of shielding electrons that surround the nucleus.
IF YOU WANT TO LEARN THIS READ BELOW.
The effective nuclear charge (Zeff) is calculated by knowing the atomic number of the atom (Z, number of protons), and the number of electrons that lie in shells closer to the nucleus than the electron you're looking at. This second part is
the shielding (S) that the electron of interest experiences. The overall calculation is: Zeff = Z - S. Determining Z is easy--just look up the atomic number of the element (it's on the periodic table). Figuring out S is also easy. The value of S is equal to the number of electrons that lie in shells with n values smaller than the n for the electron you're interested in. So, if you are calculating the shielding for a 3p electron, then all electrons with n values of 1 or 2 contribute to the shielding; this would be 2 (n=1) + 8 (n=2) = 10 total electrons, or S = 10.
Explain the basis for your order.
The shielding of electrons in the n=3 shell by 1s and 2s core electrons in these elements is approximately equal, so the effective nuclear charge increases as Z increases.
2a) How do the sizes of atoms change as we move from left to right across a row in the periodic table?
Size decreases
b) How do the sizes of atoms change as we move from top to bottom in a group in the periodic table?
Size increases
c) Arrange the following atoms in order of increasing atomic radius: C, I, Rb, and F. (Use the appropriate <, =, or > symbol to separate substances in the list.) Refer to 2a) and 2b)
F < C < I < Rb
3a) Among the nonmetallic elements, the change in atomic radius in moving one place left or right in a row is smaller than the change in moving one row up or down. Explain these observations.
A unit change in principal quantum number has a much larger effect on the size of the outermost orbital than the change in electrostatic attraction between the outer electrons and a nucleus with one more or one less proton
4 a,b,c) Refer to 2a) and 2b)
5) Write equations that show the processes that describe the first, second, and third ionization energies of a boron atom. (Omit states-of-matter in your answer.)
I1 = B --> B^+ + e^-
I2 = B^+ --> B^2+ + e^-
I3 = B^2+ --> B^3+ + e^-
6) For each of the following pairs, indicate which element has the larger first ionization energy.
Ionization energy increases going up and across (from left to right).
7) For each of the following pairs, which element will have the greater metallic character?
Use periodic table: more metallic to the left and lower down basically, as this makes it easier to form positive ions - the characterisitic of a good metal
1) Sb or As As is less metallic (higher up then Sb, both metalloids though)
2) Be or Na Be (higher and further to the right than Na, but both metals)
3) Si or P P (futher to right P is non metal Si is metalloid)
4) Cs or Au Au (further to right, but both metals)
Solid and Liquids - Phase Changes
1) Select the phase transition in each of the following situations, and indicate whether it is exothermic or endothermic.
(a) Bromine vapor turns to bromine liquid as it is cooled. condensation, exothermic
(b) Crystals of iodine disappear from an evaporating dish as they stand in a fume hood. sublimation, endothermic
(c) Rubbing alcohol in an open container slowly disappears. vaporization, endothermic
(d) Molten lava from a volcano turns into solid rock. freezing, exothermic
2) For many years, drinking water has been cooled in hot climates by evaporating it from the surfaces of canvas bags or porous clay pots. How many grams of water can be cooled from 33°C to 20.°C by the evaporation of 65 g of water? (The heat
of vaporization of water in this temperature range is 2.4 kJ/g. The specific heat of water is 4.18 J/g-K.)
m = mv · Δhv / ( m · cp ·ΔT)
= mv · Δhv / (cp ·ΔT)
= 65g · 2400J/g / [4.18J/gK · (33-20)K ] = 2870.8g
3) The fluorocarbon compound C2Cl3F3 has a normal boiling point of 47.6°C. The specific heats of C2Cl3F3(l) and
C2Cl3F3(g) are 0.91 J/g-K and 0.67 J/g-K, respectively. The heat of vaporization for the compound is 27.49 kJ/mol. Calculate the heat required to convert 22.1g of C2Cl3F3 (l) from a liquid at 4.82 C to a gas at 82.55 C
∆H = (m Cp ∆T) liquid + m ∆Hfus + (m Cp ∆T) gas
∆H = {22.1 g*(0.91 J/gC)*(47.6C - 4.82C)} + {22.1g*(1 mole / 187.4g)*(27490 J/mole)} + {22.1 g*(0.67
J/gC)*(82.55C - 47.60C)}
∆H = 860 J + 3242 J + 518 J = 4620 J = 4.62 kJ
4) For each of the following pairs of substances, predict which will have the higher melting point.
(a) Br2 or NaBr
(b)MgF2 or LiF
(c) NaCl or CO
(d) Xe or Kr
(c) C (graphite) or CH4
(d)KBr or Br2
5) The graph below represents a heating curve where the substance at low temperature is a solid.
- Which area (a, b, c) represents the location of a phase change? b
- At area labeled "b" what is the physical state of the substance (select one or more)? liquid, gas
- At area labeled "c" what is the physical state of the substance (select one or more)? liquid
6) How many kilojoules of heat is required to completely covert 85.0 grams of water at 42.0° C to steam at 100°C?
For water: s = 4.179 J/g °C Hfusion = 6.01 kJ/mol Hvap = 40.7 kJ/mol
To raise the water from its initial temperature to its boiling point:
q = cmdT
q = 4.184 * 85 * (100 - 42) = 20630 J
To vaporize the water:
85 g water (1 mol / 18.01 g) = 4.72 mol
q = m * Hv
q = 4.72 * 40700 = 192000 J
Total energy: 192000 + 20630 = 212000 = 212 kJ
7) Which of the following statements describes what can happen energetically during a phase change?
- A phase change involves the formation of molecular attractions, which releases energy.
- A phase change, for example, from liquid to vapor, requires the absorption of energy; the reverse releases this energy
- A phase change involves the breaking of molecular attractions, which requires energy.
- The energy involved in the phase change is termed enthalpy
8) a) C3H8 dispersion forces
b) SOBr2 dispersion forces, dipole-dipole forces
9) What type of intermolecular forces would you expect each of the following molecules to exhibit? (Select all that apply.)
a) CH3CH3 - dispersion forces
b) HOH - dipole-dipole interactions, dispersion forces, hydrogen bonding
c) NF3 - dipole-dipole interactions, dispersion forces
(a) Bromine vapor turns to bromine liquid as it is cooled. condensation, exothermic
(b) Crystals of iodine disappear from an evaporating dish as they stand in a fume hood. sublimation, endothermic
(c) Rubbing alcohol in an open container slowly disappears. vaporization, endothermic
(d) Molten lava from a volcano turns into solid rock. freezing, exothermic
2) For many years, drinking water has been cooled in hot climates by evaporating it from the surfaces of canvas bags or porous clay pots. How many grams of water can be cooled from 33°C to 20.°C by the evaporation of 65 g of water? (The heat
of vaporization of water in this temperature range is 2.4 kJ/g. The specific heat of water is 4.18 J/g-K.)
m = mv · Δhv / ( m · cp ·ΔT)
= mv · Δhv / (cp ·ΔT)
= 65g · 2400J/g / [4.18J/gK · (33-20)K ] = 2870.8g
3) The fluorocarbon compound C2Cl3F3 has a normal boiling point of 47.6°C. The specific heats of C2Cl3F3(l) and
C2Cl3F3(g) are 0.91 J/g-K and 0.67 J/g-K, respectively. The heat of vaporization for the compound is 27.49 kJ/mol. Calculate the heat required to convert 22.1g of C2Cl3F3 (l) from a liquid at 4.82 C to a gas at 82.55 C
∆H = (m Cp ∆T) liquid + m ∆Hfus + (m Cp ∆T) gas
∆H = {22.1 g*(0.91 J/gC)*(47.6C - 4.82C)} + {22.1g*(1 mole / 187.4g)*(27490 J/mole)} + {22.1 g*(0.67
J/gC)*(82.55C - 47.60C)}
∆H = 860 J + 3242 J + 518 J = 4620 J = 4.62 kJ
4) For each of the following pairs of substances, predict which will have the higher melting point.
(a) Br2 or NaBr
(b)MgF2 or LiF
(c) NaCl or CO
(d) Xe or Kr
(c) C (graphite) or CH4
(d)KBr or Br2
5) The graph below represents a heating curve where the substance at low temperature is a solid.
- Which area (a, b, c) represents the location of a phase change? b
- At area labeled "b" what is the physical state of the substance (select one or more)? liquid, gas
- At area labeled "c" what is the physical state of the substance (select one or more)? liquid
6) How many kilojoules of heat is required to completely covert 85.0 grams of water at 42.0° C to steam at 100°C?
For water: s = 4.179 J/g °C Hfusion = 6.01 kJ/mol Hvap = 40.7 kJ/mol
To raise the water from its initial temperature to its boiling point:
q = cmdT
q = 4.184 * 85 * (100 - 42) = 20630 J
To vaporize the water:
85 g water (1 mol / 18.01 g) = 4.72 mol
q = m * Hv
q = 4.72 * 40700 = 192000 J
Total energy: 192000 + 20630 = 212000 = 212 kJ
7) Which of the following statements describes what can happen energetically during a phase change?
- A phase change involves the formation of molecular attractions, which releases energy.
- A phase change, for example, from liquid to vapor, requires the absorption of energy; the reverse releases this energy
- A phase change involves the breaking of molecular attractions, which requires energy.
- The energy involved in the phase change is termed enthalpy
8) a) C3H8 dispersion forces
b) SOBr2 dispersion forces, dipole-dipole forces
9) What type of intermolecular forces would you expect each of the following molecules to exhibit? (Select all that apply.)
a) CH3CH3 - dispersion forces
b) HOH - dipole-dipole interactions, dispersion forces, hydrogen bonding
c) NF3 - dipole-dipole interactions, dispersion forces
S, L, Sol'n - Phase Diagrams, BP, FP
1) List the following aqueous solutions in the order of increasing boiling point: LiBr < glucose < Zn(NO3)2
2) Using data from this table, calculate the freezing and boiling points of each of the following solutions.
a) 0.40 m glucose in ethanol
Given: Kf is 1.99
normal freezing is -114.6
Kb is 1.22
boiling is 78.4
∆T = (1.99) * (0.40m) *(1)
∆T = 0.796
Normal freezing is -114.6
So the new freezing point would be -114.6 - (0.796) = -115.4°C
For boiling point: (∆)Tb = Kb*m
Kb for ethanol = 1.22
m = 0.4
(∆)Tb = 0.488
b. p . of solution = normal b. p. of water - (∆)Tb
normal b.p. of ethanol = 78.4
(∆)Tb = 0.488
b. p . of solution= 77.9°C
b) 15.0 g of decane, C10H22, in 39.6 g CHCl3
molality = mol decane / kg CHCl3
15.0 g decane x (1 mol / 142 g) = 0.106 mol
molality = 0.106 mol / 0.0396 kg = 2.67 molal
∆Tb = Kb x molality = 3.63°C/molal x 2.67 molal = 9.68°C
B.P is raised 9.68°C
B.P. = 61.2 + 9.68 = 70.88°C
∆Tf = Kf x molality = 4.68°C/molal x 2.67 molal = 12.5°C
F.P. is lowered by 12.5 C
F.P = -63.5 - 12.5 = -76.0°C
c) 0.54mol ethylene glycol and 0.12 mol KBr in 112 g H2O
KBr is a strong electrolyte : we get 0.12 mol K+ and 0.12 mol Br-
m = 0.54 + 0.12 + 0.12 / 0.112 Kg =7.0 ( at 2 significant figures)
∆T = 7 x 0.51 =3.6
boiling point = 100 + 3.6 = 103.6°C
∆T = 7 x 1.86=13
freezing point = 0 - 13 = - 13°C
3) Adrenaline is the hormone that triggers the release of extra glucose molecules in times of stress or emergency. A solution of 0.64 g of adrenaline in 36.0 g of CCl4 elevates the boiling point by 0.49°C. Calculate the molar mass of adrenaline.
Kb of CCl4 = 5.05
0.49 = 5.05 x m
m = 0.0970 = moles
adrenaline / 0.0360 Kg
moles adrenaline = 0.00349
molar mass = 0.64 g / 0.00349 = 183.2 g/mol
2) Using data from this table, calculate the freezing and boiling points of each of the following solutions.
a) 0.40 m glucose in ethanol
Given: Kf is 1.99
normal freezing is -114.6
Kb is 1.22
boiling is 78.4
∆T = (1.99) * (0.40m) *(1)
∆T = 0.796
Normal freezing is -114.6
So the new freezing point would be -114.6 - (0.796) = -115.4°C
For boiling point: (∆)Tb = Kb*m
Kb for ethanol = 1.22
m = 0.4
(∆)Tb = 0.488
b. p . of solution = normal b. p. of water - (∆)Tb
normal b.p. of ethanol = 78.4
(∆)Tb = 0.488
b. p . of solution= 77.9°C
b) 15.0 g of decane, C10H22, in 39.6 g CHCl3
molality = mol decane / kg CHCl3
15.0 g decane x (1 mol / 142 g) = 0.106 mol
molality = 0.106 mol / 0.0396 kg = 2.67 molal
∆Tb = Kb x molality = 3.63°C/molal x 2.67 molal = 9.68°C
B.P is raised 9.68°C
B.P. = 61.2 + 9.68 = 70.88°C
∆Tf = Kf x molality = 4.68°C/molal x 2.67 molal = 12.5°C
F.P. is lowered by 12.5 C
F.P = -63.5 - 12.5 = -76.0°C
c) 0.54mol ethylene glycol and 0.12 mol KBr in 112 g H2O
KBr is a strong electrolyte : we get 0.12 mol K+ and 0.12 mol Br-
m = 0.54 + 0.12 + 0.12 / 0.112 Kg =7.0 ( at 2 significant figures)
∆T = 7 x 0.51 =3.6
boiling point = 100 + 3.6 = 103.6°C
∆T = 7 x 1.86=13
freezing point = 0 - 13 = - 13°C
3) Adrenaline is the hormone that triggers the release of extra glucose molecules in times of stress or emergency. A solution of 0.64 g of adrenaline in 36.0 g of CCl4 elevates the boiling point by 0.49°C. Calculate the molar mass of adrenaline.
Kb of CCl4 = 5.05
0.49 = 5.05 x m
m = 0.0970 = moles
adrenaline / 0.0360 Kg
moles adrenaline = 0.00349
molar mass = 0.64 g / 0.00349 = 183.2 g/mol
Pressure, Colligative Prop
1 a) Calculate the mass percentage of Na2SO4 in a solution containing 12 g Na2SO4 in 400 g water
(12/400) x 100 = 3%
b) An ore contains 2.86 g of silver per ton of ore. What is the concentration of silver in ppm?
2000 lbs is 907000 grams. 2.86/907000 = 0.00000315 Convert to ppm by multiplying by 1000000: 3.15 ppm
2) A solution is made containing 14.6 g of CH3OH in 184 g H2O.
The molar mass for CH3OH = 32.0 g/mole, calculated using the atomic weights for C, H, and O.
14.6 g CH3OH x (1 mole CH3OH / 32.0 g CH3OH) = 0.456 moles CH3OH
184 g H2O x (1 mole H2O / 18.0 g H2O) = 10.2 moles H2O.
total moles = 0.456 + 10.2 = 10.7 moles
mole fraction CH3OH = (moles CH3OH / total moles) = 0.456 / 10.7 = 0.0426
mass % CH3OH = (mass of CH3OH / total mass of solution) x 100
= (14.6 g / (14.6 g + 184 g)) = 7.35%
14.6 g M/184 gH2O x 1000 gH2O/1 kg H2O x 1 mol M/32 g M = 2.48 m
3) What is the molarity of each of the following solutions? Molarity = moles per liter
a) 24.0 g Al2(SO4) in 0.350 L solution
Al2(SO4)3 has a molecular mass of 342 g
24.0 g/342 g=.70 mol
.70 mol/.350L = .2M
b) 5.35 g Mn(NO3)2 * 2H2O in 175 mL of solution
Molar mass Mn(NO3)2.2H2O = 214.9787 g/mol
Mass of Mn(NO3)2 = 178.9481/214.9787 * 5.35 = 4.453g Mn(NO3)2
Mol Mn(NO3)2 = 4.453/178.9481 = 0.0249 = 0.025 mol
Molarity = mol solute / volume in L = 0.025/0.175 = 0.142M
c) 35.0 mL of 9.00 M H2SO4 diluted to .500 L
V1C1=V2C2
35.0ml(9.00M)=C2(500mL)
(35)(9)/(500)= 0.630M
4) Calculate the number of moles of solute present in each of the following aqueous solutions.
a) 540 mL of 0.250 M SrBr2 .54*.250
b) 80 g of 0.160 m KCl
80 g = .08 kg
.08*.16
c) 126.0 g of a solution that is 6.65% glucose (C6H12O6 ) by mass?
6.65 = mass glucose x 100/ 126.0
mass glucose = 8.38 g
moles = 8.38 g/180 g/mol = 0.0466
5)
a)Calculate the vapor pressure of water above a solution prepared by dissolving 39.5 g in glycerin (C3H8O3) to 149 g of water at 343 K
343 - 273 = 70 °C
vapor pressure water at 70 °C = 233.7 torr
moles glycerin = 39.5 g /92.097 g/mol=0.420
moles water = 149 g/ 18.02 g/mol=8.27
mole fraction water = 8.27/ 8.27 + 0.420=0.952
p = 0.952 x 233.7=222.5 torr
1.00 x 10^2 - 10.5 =89.5 torr
89.5 = X 1.00 x 10^2
X = mole fraction solvent = 0.895
moles ethanol = 1000 g/46.069
g/mol=21.7
0.895 = 21.7 / 21.7 + x
b) Calculate the mass of ethylene glycol (C2H6O2) that must be added to 1.00 kg of ethanol (C2H5OH) to
reduce its vapor pressure by 10.5 torr at 35°C. The vapor pressure of pure ethanol at 35°C is 1.00 multiplied by 102 torr.
19.4 + 0.895 x = 21.7
x = 2.57 = moles C2H6O2
mass C2H6O2 = 2.57 mol x
62.07 g/mol = 159.5 g
6) List the following aqueous solutions in the order of increasing boiling point:
0.050 m LiBr < 0.120 m glucose < 0.050 m Zn(NO3)2
7) What is the freezing point of an aqueous solution that boils at 107.5°C?
Kb = ebullioscopic constant (aka boiling point elevation constant) for the solvent (water in this case) = 0.512 C/m
Kf = cryoscopic constant (aka freezing point depression constant) for the solvent. = 1.86 C / m for water
∆Tf = (107.5 C - 100 C) x (1.86 C/m) / (0.512 C/m) = 27.2°C
8) Seawater contains 3.4 g of salts for every liter of solution. Assuming that the solute consists entirely of NaCl (over 90% is), calculate the osmotic pressure of seawater at 20°C.
moles NaCl = 3.4 g / 58.4428 g/mol= 0.058
molarity = 0.058 mol / 1 L = 0.058
i = Vant'Hoff factor = 2
osmotic pressure = 0.058 x 2 x 0.0821 x 293 K = 2.8 atm
9) A dilute aqueous solution of an organic compound soluble in water is formed by dissolving 2.48 g of the compound in water to form 0.250 L solution. The resulting solution has an osmotic pressure of 0.627 atm at 25°C. Assuming that
the organic compound is a nonelectrolyte, what is its molar mass?
p = 0.627atm, 298K = t
again, π = iMRT, this is a non-electrolyte so the van't hoff factor = 1
0.627atm = M x 0.0821L-atm/mole-K x 298K
M = 0.026M
(2.48g / ?g/mole) / 0.25L = 0.026M
mass = 381.54 g/mol
10) Guess and check ;)
11) The Henry's law constant for helium gas in water at 30°C is 3.7 10-4 M/atm and the constant for N2 at 30°C is6.0 10-4 M/atm. If the two gases are each present at .65 atm pressure, calculate the solubility of each gas.
a) He
c=kp
c=0.65*3.7*10^-4= 2.4*10^-4M
b) N2
c=kp
c=0.65*6*10^-4= 3.9*10^-4M
(12/400) x 100 = 3%
b) An ore contains 2.86 g of silver per ton of ore. What is the concentration of silver in ppm?
2000 lbs is 907000 grams. 2.86/907000 = 0.00000315 Convert to ppm by multiplying by 1000000: 3.15 ppm
2) A solution is made containing 14.6 g of CH3OH in 184 g H2O.
The molar mass for CH3OH = 32.0 g/mole, calculated using the atomic weights for C, H, and O.
14.6 g CH3OH x (1 mole CH3OH / 32.0 g CH3OH) = 0.456 moles CH3OH
184 g H2O x (1 mole H2O / 18.0 g H2O) = 10.2 moles H2O.
total moles = 0.456 + 10.2 = 10.7 moles
mole fraction CH3OH = (moles CH3OH / total moles) = 0.456 / 10.7 = 0.0426
mass % CH3OH = (mass of CH3OH / total mass of solution) x 100
= (14.6 g / (14.6 g + 184 g)) = 7.35%
14.6 g M/184 gH2O x 1000 gH2O/1 kg H2O x 1 mol M/32 g M = 2.48 m
3) What is the molarity of each of the following solutions? Molarity = moles per liter
a) 24.0 g Al2(SO4) in 0.350 L solution
Al2(SO4)3 has a molecular mass of 342 g
24.0 g/342 g=.70 mol
.70 mol/.350L = .2M
b) 5.35 g Mn(NO3)2 * 2H2O in 175 mL of solution
Molar mass Mn(NO3)2.2H2O = 214.9787 g/mol
Mass of Mn(NO3)2 = 178.9481/214.9787 * 5.35 = 4.453g Mn(NO3)2
Mol Mn(NO3)2 = 4.453/178.9481 = 0.0249 = 0.025 mol
Molarity = mol solute / volume in L = 0.025/0.175 = 0.142M
c) 35.0 mL of 9.00 M H2SO4 diluted to .500 L
V1C1=V2C2
35.0ml(9.00M)=C2(500mL)
(35)(9)/(500)= 0.630M
4) Calculate the number of moles of solute present in each of the following aqueous solutions.
a) 540 mL of 0.250 M SrBr2 .54*.250
b) 80 g of 0.160 m KCl
80 g = .08 kg
.08*.16
c) 126.0 g of a solution that is 6.65% glucose (C6H12O6 ) by mass?
6.65 = mass glucose x 100/ 126.0
mass glucose = 8.38 g
moles = 8.38 g/180 g/mol = 0.0466
5)
a)Calculate the vapor pressure of water above a solution prepared by dissolving 39.5 g in glycerin (C3H8O3) to 149 g of water at 343 K
343 - 273 = 70 °C
vapor pressure water at 70 °C = 233.7 torr
moles glycerin = 39.5 g /92.097 g/mol=0.420
moles water = 149 g/ 18.02 g/mol=8.27
mole fraction water = 8.27/ 8.27 + 0.420=0.952
p = 0.952 x 233.7=222.5 torr
1.00 x 10^2 - 10.5 =89.5 torr
89.5 = X 1.00 x 10^2
X = mole fraction solvent = 0.895
moles ethanol = 1000 g/46.069
g/mol=21.7
0.895 = 21.7 / 21.7 + x
b) Calculate the mass of ethylene glycol (C2H6O2) that must be added to 1.00 kg of ethanol (C2H5OH) to
reduce its vapor pressure by 10.5 torr at 35°C. The vapor pressure of pure ethanol at 35°C is 1.00 multiplied by 102 torr.
19.4 + 0.895 x = 21.7
x = 2.57 = moles C2H6O2
mass C2H6O2 = 2.57 mol x
62.07 g/mol = 159.5 g
6) List the following aqueous solutions in the order of increasing boiling point:
0.050 m LiBr < 0.120 m glucose < 0.050 m Zn(NO3)2
7) What is the freezing point of an aqueous solution that boils at 107.5°C?
Kb = ebullioscopic constant (aka boiling point elevation constant) for the solvent (water in this case) = 0.512 C/m
Kf = cryoscopic constant (aka freezing point depression constant) for the solvent. = 1.86 C / m for water
∆Tf = (107.5 C - 100 C) x (1.86 C/m) / (0.512 C/m) = 27.2°C
8) Seawater contains 3.4 g of salts for every liter of solution. Assuming that the solute consists entirely of NaCl (over 90% is), calculate the osmotic pressure of seawater at 20°C.
moles NaCl = 3.4 g / 58.4428 g/mol= 0.058
molarity = 0.058 mol / 1 L = 0.058
i = Vant'Hoff factor = 2
osmotic pressure = 0.058 x 2 x 0.0821 x 293 K = 2.8 atm
9) A dilute aqueous solution of an organic compound soluble in water is formed by dissolving 2.48 g of the compound in water to form 0.250 L solution. The resulting solution has an osmotic pressure of 0.627 atm at 25°C. Assuming that
the organic compound is a nonelectrolyte, what is its molar mass?
p = 0.627atm, 298K = t
again, π = iMRT, this is a non-electrolyte so the van't hoff factor = 1
0.627atm = M x 0.0821L-atm/mole-K x 298K
M = 0.026M
(2.48g / ?g/mole) / 0.25L = 0.026M
mass = 381.54 g/mol
10) Guess and check ;)
11) The Henry's law constant for helium gas in water at 30°C is 3.7 10-4 M/atm and the constant for N2 at 30°C is6.0 10-4 M/atm. If the two gases are each present at .65 atm pressure, calculate the solubility of each gas.
a) He
c=kp
c=0.65*3.7*10^-4= 2.4*10^-4M
b) N2
c=kp
c=0.65*6*10^-4= 3.9*10^-4M
Kinetics, Ch 14, WA #1
The following data were collected for the rate of disappearance of NO in the reaction 2 NO(g) + O2(g) → 2 NO2(g).
(a) What is the rate law for the reaction? (Rate equations take the general form 'rate = k . [A] . [B]'.)
rate = k[NO]^2[O_2]
(b) What are the units of the rate constant? M-2·s-1
(c) What is the average value of the rate constant calculated from the three data sets? 7105.064
(d) What is the rate of disappearance of NO when [NO] = 0.0750 M and [O2] = 0.0100 M? .3997 M/s
(e) What is the rate of disappearance of O2 at the concentrations given in part (d)? .1998 M/s
(a) What is the rate law for the reaction? (Rate equations take the general form 'rate = k . [A] . [B]'.)
rate = k[NO]^2[O_2]
(b) What are the units of the rate constant? M-2·s-1
(c) What is the average value of the rate constant calculated from the three data sets? 7105.064
(d) What is the rate of disappearance of NO when [NO] = 0.0750 M and [O2] = 0.0100 M? .3997 M/s
(e) What is the rate of disappearance of O2 at the concentrations given in part (d)? .1998 M/s
Kinetics, Ch 14, WA #2
1) The following data were measured for the reaction given below. BF3(g) + NH3(g) → F3BNH3(g)
(a) What is the rate law for the reaction? k[BF3][NH3]
(b) What is the overall order of the reaction? second order
(c) What is the value of the rate constant for the reaction? 3.41 M -1·s-1
(d) What is the rate when [BF3] = 0.100 M and [NH3] = 0.500 M? .170 M/s
2) The decomposition of N2O5 in carbon tetrachloride proceeds as follows. 2 N2O5 → 4 NO2 + O2The rate law is first order in N2O5. At 64°C the rate constant, k, is 4.82 10-3 s-1.
(a) Write the rate law for the reaction: rate = k[N_2O_5]
(b) What is the rate of reaction when [N2O5] = 0.0120 M? rate = k[N2O5] = (4.82 x 10-3) x 0.0120 = 5.784e-5
(c) What happens to the rate when the concentration of N2O5 is doubled to 0.0240 M? place these values in the rate equation taken in (a) and you will see that rate also doubles. 2*5.784e-5
3)
a) Consider the combustion of ethylene, given below. C2H4(g) + 3 O2(g) → 2 CO2 (g) + 2 H2O (g)
If the concentration of C2H4 is decreasing at the rate of 0.22 M/s, what are the rates of change in the concentrations of CO2 and H2O?
CO2: .22*2 M/s
H2O: .22*2 M/s
b) The rate of decrease in N2H4 partial pressure in a closed reaction vessel from the following reaction is 50 torr/hr. N2H4 (g) + H2 (g) → 2 NH3 (g)
What are the rates of change of NH3 partial pressure and total pressure in the vessel?
NH3: 2*50 torr/hr
total pressure: 0 torr/hr
(a) What is the rate law for the reaction? k[BF3][NH3]
(b) What is the overall order of the reaction? second order
(c) What is the value of the rate constant for the reaction? 3.41 M -1·s-1
(d) What is the rate when [BF3] = 0.100 M and [NH3] = 0.500 M? .170 M/s
2) The decomposition of N2O5 in carbon tetrachloride proceeds as follows. 2 N2O5 → 4 NO2 + O2The rate law is first order in N2O5. At 64°C the rate constant, k, is 4.82 10-3 s-1.
(a) Write the rate law for the reaction: rate = k[N_2O_5]
(b) What is the rate of reaction when [N2O5] = 0.0120 M? rate = k[N2O5] = (4.82 x 10-3) x 0.0120 = 5.784e-5
(c) What happens to the rate when the concentration of N2O5 is doubled to 0.0240 M? place these values in the rate equation taken in (a) and you will see that rate also doubles. 2*5.784e-5
3)
a) Consider the combustion of ethylene, given below. C2H4(g) + 3 O2(g) → 2 CO2 (g) + 2 H2O (g)
If the concentration of C2H4 is decreasing at the rate of 0.22 M/s, what are the rates of change in the concentrations of CO2 and H2O?
CO2: .22*2 M/s
H2O: .22*2 M/s
b) The rate of decrease in N2H4 partial pressure in a closed reaction vessel from the following reaction is 50 torr/hr. N2H4 (g) + H2 (g) → 2 NH3 (g)
What are the rates of change of NH3 partial pressure and total pressure in the vessel?
NH3: 2*50 torr/hr
total pressure: 0 torr/hr
Kinetics, Ch 14, WA #3
1) The decomposition of sulfuryl chloride (SO2Cl2) is a first-order process. The rate constant for the decomposition at 660 K is 4.5 10-2 s-1.
(a) If we begin with an initial SO2Cl2 pressure of 335 torr, what is the pressure of this substance after 71 s?
ln x / 335 = - 4.5 x 10^-2 x 71 = - 3.195
e ^ - 3.195 = 0.0410
x / 335 = 0.0410
x = 13.7 torr
(b) At what time will the pressure of SO2Cl2 decline to one eighth its initial value?
335/8=41.9
ln 41.9 / 335 = - 4.5 x 10^-2t
- 2.08 = - 4.5 x 10^-2 t
t = 46.2 s
2) This reaction has the rate constant k = 0.63 M -1 s-1.
2 NO2 → 2 NO + O2
Based on the units for k, is this reaction first or second order in NO2?
second order
If the initial concentration of NO2 is 0.133 M, how long would it take for the concentration to decrease to 0.020 M?
Integrated second order rate equation is:
(1/[A]) - (1/[A]o) = kt
t = (1/[A] - 1/[A]o) / k
t = (1/0.020 - 1/0.133) / 0.63 1/Ms
t = 67.4 s
(a) If we begin with an initial SO2Cl2 pressure of 335 torr, what is the pressure of this substance after 71 s?
ln x / 335 = - 4.5 x 10^-2 x 71 = - 3.195
e ^ - 3.195 = 0.0410
x / 335 = 0.0410
x = 13.7 torr
(b) At what time will the pressure of SO2Cl2 decline to one eighth its initial value?
335/8=41.9
ln 41.9 / 335 = - 4.5 x 10^-2t
- 2.08 = - 4.5 x 10^-2 t
t = 46.2 s
2) This reaction has the rate constant k = 0.63 M -1 s-1.
2 NO2 → 2 NO + O2
Based on the units for k, is this reaction first or second order in NO2?
second order
If the initial concentration of NO2 is 0.133 M, how long would it take for the concentration to decrease to 0.020 M?
Integrated second order rate equation is:
(1/[A]) - (1/[A]o) = kt
t = (1/[A] - 1/[A]o) / k
t = (1/0.020 - 1/0.133) / 0.63 1/Ms
t = 67.4 s
Kinetics, Ch 14, WA #4
1) Molecular iodine, I2(g), dissociates into iodine atoms at 625 K with a first-order rate constant of 0.271 s-1.
a) 2.56 s
b) If you start with 0.052 M I2 at this temperature, how much will remain after 7.68 s assuming that the iodine atoms do not recombine to form I2?
ln[A] = - k t + ln[Ao], where [A] is the concentration after time = t, k is the rate constant, and [Ao] is the original concentration.
ln[A] = - (0.271 1/s)(7.68 s) + ln (0.052)
ln[A] = - 2.08 + (-2.96) = - 5.04
[A] = e^-5.04 = 0.0065 M
2) The first-order rate constant for the decomposition of N2O5, given below, at 70°C is 6.82 10-3 s-1. Suppose we start with 0.0400 mol N2O5(g) in a volume of 4.0 L.
2 N2O5(g) → 4 NO2(g) + O2(g)
a) How many moles of N2O5 will remain after 1.5 min?
1st order reaction formula : Ln(Af/Ao) = -kt
rearrange to solve for the ratio (Af/Ao) = e^(-kt)
time converted to seconds: t = (1.5 min) x [(60 s)/(1 min)] = 90 s
(Af/Ao) = e^[-(6.82x10^-3 s-1)(90 s)]
(Af/Ao) = e^[-6.12x10^-1] = 0.54
Af = 0.54Ao
Af = 0.54(0.0400 moles) = 0.022 moles
b) How many minutes will it take for the quantity of N2O5 to drop to 0.005 mol?
Plug values into rate equation:
Ln(0.005/0.0400) = -(6.82x10^-3 s-1)t
- 2.079 = -(6.82x10^-3 s-1)t
t = 305 s /60 = 5.083 min
c) What is the half-life of N2O5 at 70°C?
t(1/2) = 0.693/k
t(1/2) = 0.693/(6.82x10^-3 s-1) = 102 s /60 = 1.7 min
a) 2.56 s
b) If you start with 0.052 M I2 at this temperature, how much will remain after 7.68 s assuming that the iodine atoms do not recombine to form I2?
ln[A] = - k t + ln[Ao], where [A] is the concentration after time = t, k is the rate constant, and [Ao] is the original concentration.
ln[A] = - (0.271 1/s)(7.68 s) + ln (0.052)
ln[A] = - 2.08 + (-2.96) = - 5.04
[A] = e^-5.04 = 0.0065 M
2) The first-order rate constant for the decomposition of N2O5, given below, at 70°C is 6.82 10-3 s-1. Suppose we start with 0.0400 mol N2O5(g) in a volume of 4.0 L.
2 N2O5(g) → 4 NO2(g) + O2(g)
a) How many moles of N2O5 will remain after 1.5 min?
1st order reaction formula : Ln(Af/Ao) = -kt
rearrange to solve for the ratio (Af/Ao) = e^(-kt)
time converted to seconds: t = (1.5 min) x [(60 s)/(1 min)] = 90 s
(Af/Ao) = e^[-(6.82x10^-3 s-1)(90 s)]
(Af/Ao) = e^[-6.12x10^-1] = 0.54
Af = 0.54Ao
Af = 0.54(0.0400 moles) = 0.022 moles
b) How many minutes will it take for the quantity of N2O5 to drop to 0.005 mol?
Plug values into rate equation:
Ln(0.005/0.0400) = -(6.82x10^-3 s-1)t
- 2.079 = -(6.82x10^-3 s-1)t
t = 305 s /60 = 5.083 min
c) What is the half-life of N2O5 at 70°C?
t(1/2) = 0.693/k
t(1/2) = 0.693/(6.82x10^-3 s-1) = 102 s /60 = 1.7 min
Equilibrium, Ch 15, #1
1 a) In the reaction below, what does the symbol "" mean? OH-(aq) + NH4+ (aq) H2O (l) + NH3 (g) Note: check all that apply.
The reaction is reversible. No net changes in concentrations are occuring. The rate of forward and reverse reactions are the same. If we knew the concentrations of the species, we could determine an equilibrium constant value and the extent of the reaction.
b) The equilibrium constant for a reaction is equal to: the product of the products raised to their stoichiometric coefficents divided by the product of the reactants raised to their stoichiometric coefficents.
c) Identify the equilibrium constant expression in terms of the concentrations of the reactants and products for the following reaction: 2HNO2(aq) + PO43-(aq) 2NO21-(aq) + H2PO41-(aq)
K = ([NO21-]2 [H2PO41-]) / ([HNO2]2 [PO43-])
d) Identify the equilibrium constant expression in terms of the concentrations of the reactants and products for the following reaction: 3Fe(s) + 2Au3+(aq) 2Au(s) + 3Fe2+(aq)
K = [Fe2+]3 / [Au3+]2
2) Indicate whether each of the following reactions lies to the right, favoring the formation of products, or lies to the left, favoring formation of reactants.
a) 2 HI(g) H2(g) + I2(g); Keq = 1.8 10-2
If Keq = <1.00 [products] < [reactants] : left side is favoured
b) CO(g) + H2O(g) CO2(g) + H2(g); Keq = 2.3 101
IF Keq > 1.0 [products] > [reactants] : right side is favoured
3) Phosphorus trichloride gas and chlorine gas react to form phosphorus pentachloride gas. PCl3(g) + Cl2(g) PCl5(g)
A gas vessel is charged with a mixture of PCl3(g) and Cl2(g), which is allowed to equilibrate at 450 K. At equilibrium the partial pressures of the three gases are PPCl3 = 0.117atm, PCl2 = 0.198 atm, and PPCl5 = 1.55 atm.
a) What is the value of Kp at this temperature? 1.55/(.117*.198) = 66.9
b) Does the equilibrium favor reactants or products? The equilibrium favors products.
4) 2 SO2(g) + O2(g) 2 SO3(g)
An equilibrium mixture for the reaction above contains [SO2] = 1.00 M, [O2] = 2.00 M, and [SO3] = 3.50 M. What is the value of the equilibrium constant, Kc, for the reaction? [SO3]^2/{(O2)*[SO2]^2} = 6.125
5) C2H2(g) + 2 Cl2(g) C2H2Cl4(g)
An equilibrium mixture for the reaction above contains [C2H2] = 3.50 M, [Cl2] = 1.00 M, and [C2H2Cl4] = 2.50 M. What is the value of the equilibrium constant, Kc, for the reaction? [C2H2Cl4]/{(Cl2)^2*[C2H2]} = .714
6) H2O(g) + CO(g) CO2(g) + H2(g)
The equilibrium constant Kc = 4.00 for the reaction above. An equilibrium mixture contains [CO2] = 0.350 M, [H2] = 0.350 M, and [CO] = 0.150 M. What is the equilibrium [H2O]?
4.00 = [H2][CO2]/{[H2O][CO]} Solve for H2O.
[H2O] = .204
The reaction is reversible. No net changes in concentrations are occuring. The rate of forward and reverse reactions are the same. If we knew the concentrations of the species, we could determine an equilibrium constant value and the extent of the reaction.
b) The equilibrium constant for a reaction is equal to: the product of the products raised to their stoichiometric coefficents divided by the product of the reactants raised to their stoichiometric coefficents.
c) Identify the equilibrium constant expression in terms of the concentrations of the reactants and products for the following reaction: 2HNO2(aq) + PO43-(aq) 2NO21-(aq) + H2PO41-(aq)
K = ([NO21-]2 [H2PO41-]) / ([HNO2]2 [PO43-])
d) Identify the equilibrium constant expression in terms of the concentrations of the reactants and products for the following reaction: 3Fe(s) + 2Au3+(aq) 2Au(s) + 3Fe2+(aq)
K = [Fe2+]3 / [Au3+]2
2) Indicate whether each of the following reactions lies to the right, favoring the formation of products, or lies to the left, favoring formation of reactants.
a) 2 HI(g) H2(g) + I2(g); Keq = 1.8 10-2
If Keq = <1.00 [products] < [reactants] : left side is favoured
b) CO(g) + H2O(g) CO2(g) + H2(g); Keq = 2.3 101
IF Keq > 1.0 [products] > [reactants] : right side is favoured
3) Phosphorus trichloride gas and chlorine gas react to form phosphorus pentachloride gas. PCl3(g) + Cl2(g) PCl5(g)
A gas vessel is charged with a mixture of PCl3(g) and Cl2(g), which is allowed to equilibrate at 450 K. At equilibrium the partial pressures of the three gases are PPCl3 = 0.117atm, PCl2 = 0.198 atm, and PPCl5 = 1.55 atm.
a) What is the value of Kp at this temperature? 1.55/(.117*.198) = 66.9
b) Does the equilibrium favor reactants or products? The equilibrium favors products.
4) 2 SO2(g) + O2(g) 2 SO3(g)
An equilibrium mixture for the reaction above contains [SO2] = 1.00 M, [O2] = 2.00 M, and [SO3] = 3.50 M. What is the value of the equilibrium constant, Kc, for the reaction? [SO3]^2/{(O2)*[SO2]^2} = 6.125
5) C2H2(g) + 2 Cl2(g) C2H2Cl4(g)
An equilibrium mixture for the reaction above contains [C2H2] = 3.50 M, [Cl2] = 1.00 M, and [C2H2Cl4] = 2.50 M. What is the value of the equilibrium constant, Kc, for the reaction? [C2H2Cl4]/{(Cl2)^2*[C2H2]} = .714
6) H2O(g) + CO(g) CO2(g) + H2(g)
The equilibrium constant Kc = 4.00 for the reaction above. An equilibrium mixture contains [CO2] = 0.350 M, [H2] = 0.350 M, and [CO] = 0.150 M. What is the equilibrium [H2O]?
4.00 = [H2][CO2]/{[H2O][CO]} Solve for H2O.
[H2O] = .204
Equilibrium, Ch 15, #2
1) Calculate Kc at 303 K for the following equation if Kp = 34.5 at this temperature. SO2(g) + Cl2(g) SO2Cl2(g)
delta n = 1 - 1 - 1 = -1
34.5 = Kc ( 0.08206 x 303)^-1
Kc = 857.8
3)A flask is charged with 1.500 atm of N2O4 (g) and 1.00 atm NO2 (g) at 25°C, and the following equilibrium is achieved: N2O4(g) --> 2NO2(g).
After equilibrium is reached, the partial pressure of NO2 is 0.512atm.
a) What is the equilibrium partial pressure of N2O4?
1.5 .. . . . . .1.0
change
+x. . . .. . . . -2x
1.0 -2x = 0.512
2x = 0.488
x = 0.244 atm
partial pressure N2O4 = 1.5 + 0.244 = 1.744 atm
b) What's the value of Kp for the reaction? Kp = (0.512)^2 / 1.744 = 0.150
5) At 900 K the following reaction has Kp = 0.345.
2 SO2(g) + O2(g) <==> 2 SO3(g)
In an equilibrium mixture the partial pressures of SO2 and O2 are 0.135 atm and 0.455 atm, respectively. What is the equilibrium partial pressure of SO3 in the mixture?
Kp = [SO3]^2 / [SO2]^2 [O2]
0.345 = [SO3]^2 / [0.135]^2 [0.455]
0.345 = [SO3]^2 / 0.00829
[SO3]^2 = 0.00286
[SO3] = 0.0535 atm
delta n = 1 - 1 - 1 = -1
34.5 = Kc ( 0.08206 x 303)^-1
Kc = 857.8
3)A flask is charged with 1.500 atm of N2O4 (g) and 1.00 atm NO2 (g) at 25°C, and the following equilibrium is achieved: N2O4(g) --> 2NO2(g).
After equilibrium is reached, the partial pressure of NO2 is 0.512atm.
a) What is the equilibrium partial pressure of N2O4?
1.5 .. . . . . .1.0
change
+x. . . .. . . . -2x
1.0 -2x = 0.512
2x = 0.488
x = 0.244 atm
partial pressure N2O4 = 1.5 + 0.244 = 1.744 atm
b) What's the value of Kp for the reaction? Kp = (0.512)^2 / 1.744 = 0.150
5) At 900 K the following reaction has Kp = 0.345.
2 SO2(g) + O2(g) <==> 2 SO3(g)
In an equilibrium mixture the partial pressures of SO2 and O2 are 0.135 atm and 0.455 atm, respectively. What is the equilibrium partial pressure of SO3 in the mixture?
Kp = [SO3]^2 / [SO2]^2 [O2]
0.345 = [SO3]^2 / [0.135]^2 [0.455]
0.345 = [SO3]^2 / 0.00829
[SO3]^2 = 0.00286
[SO3] = 0.0535 atm